Removal of Azo dyes with Xanthan
DOI:
https://doi.org/10.29356/jmcs.v63i4.699Keywords:
Xanthan, azo dyes, removal efficiency, polyhydroxyoxoaluminum cations, aggregationAbstract
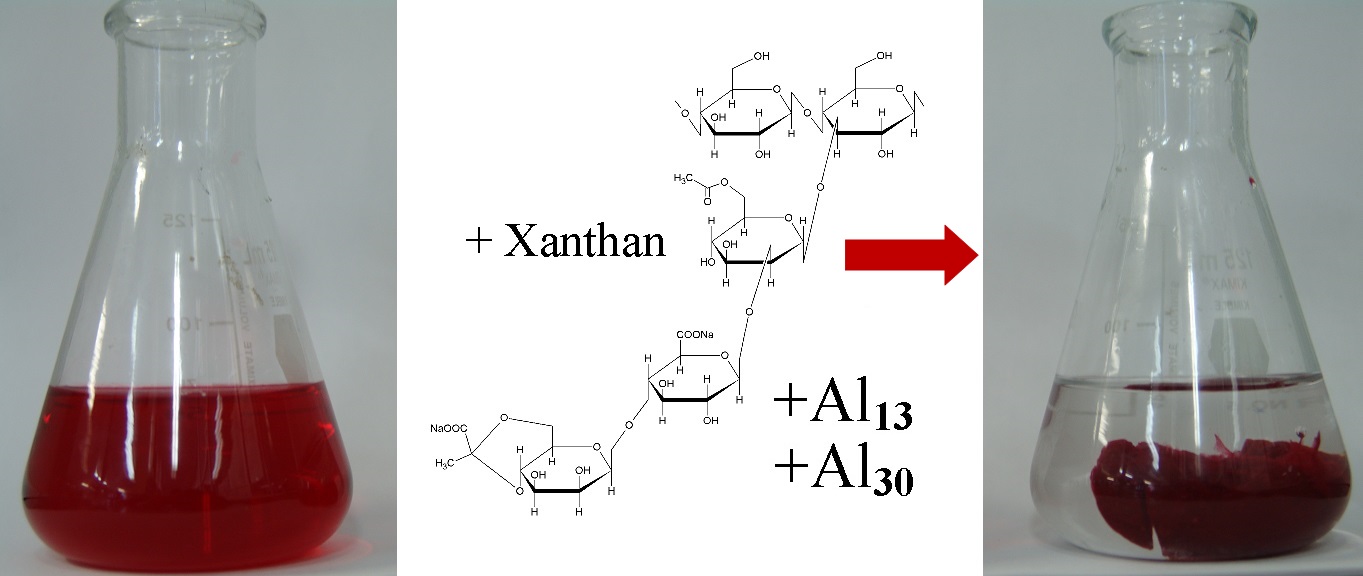
The interaction among Xanthan (XANT) and three azo dyes: Direct blue 1 (DB1), Direct red 81 (DR81), and Direct black 22 (DB22) was studied. The Xanthan-dye-Al product was formed after the addition of AlCl3 to a Xanthan-Dye adduct containing solution. It was proposed that polyhydroxyoxoaluminum clusters named CAL-13 and CAL-30 react with this adduct producing a Xanthanate aluminum network, XANT-Al, and as a consequence a decrease in dye concentration in an aqueous medium was observed. The removal efficiencies obtained were the following: DB1 (99 %), DB22 (99 %) and DR81 (94 %), demonstrating that this dye removal method is very efficient. The Zimm-Bragg model adequately described the experimental data and the order observed in the Ku (nucleation) and U (aggregation) parameters from this model was the following: DB1>DB22> DR81. Evidence suggests that physicochemical properties of dyes such as charge, molecular weight, aggregation ability and the capacity of XANT-Al to trap dye molecules are involved in the high removal values. Moreover, the dye binding mechanisms include: electrostatic, hydrogen bonding and hydrophobic interactions that determine the magnitude of the parameters Ku and U. These findings suggest that the XANT polymer is a good option to remove azo dyes from an aqueous medium.
Downloads
References
Freeman H.S.; Mock G.N., In: Handbook of industrial chemistry and Biotechnology, Kent, J.A., Ed., Springer, Boston, EUA, 2012, 475-548. DOI: 10.1007/978-1-4614-4259-2_13 DOI: https://doi.org/10.1007/978-1-4614-4259-2_13
Holme, I., in: Handbook of Technical Textiles; 2nd. ed., Vol.1, Horrocks, A. R.; Anand, S. C., Eds., Woddhead Publishing, Cambridge, 2015, 231-284. DOI: https://doi.org/10.1016/B978-1-78242-458-1.00009-1
Yagub, M. T.; Sen, T. K.; Afroze, S.; Ang, H M. Adv. Colloid Interface Sci. 2014, 209, 172–184 DOI: http://dx.doi.org/10.1016/j.cis.2014.04.002 DOI: https://doi.org/10.1016/j.cis.2014.04.002
Gulrez, S. K. H.; Al-Assafa, S.; Fang, Y.; Phillips, G. O.; Gunning, A. P. Carbohydr. Polym. 2012, 90, 1235–1243 DOI: http://dx.doi.org/10.1016/j.carbpol.2012.06.055 DOI: https://doi.org/10.1016/j.carbpol.2012.06.055
Makhado, E.; Pandey, S.; Nomngongo, P. N.; Ramontja, J. Carbohydr. Polym. 2017, 176, 315–326 DOI: http://dx.doi.org/10.1016/j.carbpol.2017.08.093 DOI: https://doi.org/10.1016/j.carbpol.2017.08.093
Mittal, H.; Parashar, V.; Mishra, S. B.; Mishra, A. K. Chem. Eng. J. 2014, 255, 471–482 DOI: http://dx.doi.org/10.1016/j.cej.2014.04.098 DOI: https://doi.org/10.1016/j.cej.2014.04.098
Ghorai, s.; Sarkar, A. K.; Panda, A. B.; Pal, S. Biores. Technol. 2013, 144, 485–491 DOI: http://dx.doi.org/10.1016/j.biortech.2013.06.108 DOI: https://doi.org/10.1016/j.biortech.2013.06.108
Ghorai, S.; Sarkar, A.; Raoufi, M.; Panda, A. B.; Schönherr, H.; Pal, S. ACS Appl. Mater. Interfaces 2014, 6, 4766?4777 DOI: http://dx.doi.org/10.1021/am4055657 DOI: https://doi.org/10.1021/am4055657
Pandey, S.; Mishra, S. V. Carbohydr. Polym. 2012, 90, 370–379. DOI: http://dx.doi.org/10.1016/j.carbpol.2012.05.053 DOI: https://doi.org/10.1016/j.carbpol.2012.05.053
Pal, A.; Majumder, K.; Bandyopadhyay, A. Carbohydr. Polym. 2016, 152, 41–50. DOI: http://dx.doi.org/10.1016/j.carbpol.2016.06.064 DOI: https://doi.org/10.1016/j.carbpol.2016.06.064
Iftekhar, S.; Srivastava, V.; Hammouda, S. B.; Sillanpääa, M. Carbohydr. Polym. 2018, 194, 274–284. DOI: https://doi.org/10.1016/j.carbpol.2018.04.054
Xin, J.; Han, J.; Zheng, X.; Shao, H.; Kolditz, O. J. Environ. Manage. 2015, 150, 420-426. DOI: http://dx.doi.org/10.1016/j.jenvman.2014.12.022 DOI: https://doi.org/10.1016/j.jenvman.2014.12.022
Chen, X.; Li, P.; Kang, Y.; Zeng, X.; Xie, Y.; Zhang, Y.; Wang, Y.; Xie, T. Carbohydr. Polym. 2019, 206, 94–101. DOI: https://doi.org/10.1016/j.carbpol.2018.10.092
Zhang, Q.; Hua, X. M.; Wu, M. Y.; Wang, M. M.; Zhao, Y. Y.; Li, T. T. React. Funct. Polym. 2019, 136, 34–43. DOI: https://doi.org/10.1016/j.reactfunctpolym.2019.01.002
Li, M and Buschle-Diller, G. Int. J. Biol. Macromol. 2017, 101, 481–489. DOI:http://dx.doi.org/10.1016/j.ijbiomac.2017.03.091 DOI: https://doi.org/10.1016/j.ijbiomac.2017.03.091
Chaudhary, S.; Sharma, J.; Kaith, B. S.; Yadav, S.; Sharma, A. K.; Goel, A. Ecotoxicol. Environ. Saf. 2018, 149, 150–158 DOI: https://doi.org/10.1016/j.ecoenv.2017.11.030
Zheng, M.; Lian, F.; Xiong, Y.; Liu, B.; Zhu, Y.; Miao, S.; Zhang, L.; Zheng, B. Food Chem. 2019, 272, 574–579. DOI: https://doi.org/10.1016/j.foodchem.2018.08.083
Sukriti; Sharma, J.; Pruthi, V.; Anand, P.; Chaddha, A. P. S.; Bhatia, J.; Kaith, B. S. RSC Adv. 2016, 6, 74300–74313. DOI: https://doi.org/10.1039/c6ra12715k
Sukriti; Sharma, J.; Chadha, A. S.; Pruthi, V.; Anand, P.; Bhatia, J.; Kaith, B. S. J. Environ. Manage. 2017, 190, 176-187. DOI: http://dx.doi.org/10.1016/j.jenvman.2016.12.065 DOI: https://doi.org/10.1016/j.jenvman.2016.12.065
Sharma, J.; Sukriti; Anand, P.; Pruthi, V.; Chaddha, A. S.; Bhatia, J.; Kaith, B. S. Mater. Chem. Phys. 2017, 196, 270-283. DOI: : http://dx.doi.org/0.1016/j.matchemphys.2017.04.042 DOI: https://doi.org/10.1016/j.matchemphys.2017.04.042
Sharma, A. K.; Priya; Kaith, B. S.; Bajaj, S.; Bhatia, J. K.; Panchal, S.; Sharma, N.; Tanwar, V. Colloid. Surface. B. 2019, 175, 314–323. DOI: https://doi.org/10.1016/j.colsurfb.2018.12.017
Mittal, H.; Kumar, v., Saruchi; Ray, S. S. Int. J. of Biol. Macromol. 2016, 89, 1–11 DOI: http://dx.doi.org/10.1016/j.ijbiomac.2016.04.050 DOI: https://doi.org/10.1016/j.ijbiomac.2016.04.050
Thakur, S.; Pandey, S.; Arotiba, O. A. Int. J. of Biol. Macromol. 2017, 103, 596–604 DOI: http://dx.doi.org/10.1016/j.ijbiomac.2017.05.087 DOI: https://doi.org/10.1016/j.ijbiomac.2017.05.087
Kaith, B. S.; Sukriti; Sharma, J.; Kaur, T.; Sethi, S.; Shanker, U.; Jassal, V. Iran. Polym. J. 2016, 25, 787–797 DOI: http://dx.doi.org/10.1007/s13726-016-0467-z DOI: https://doi.org/10.1007/s13726-016-0467-z
Makhado, E.; Pandey, S.; Ramontja, J. Int. J. Biol. Macromol. 2018, 119, 255-269. DOI: https://dx.doi.org/10.1016/j.ijbiomac.2018.07.104 DOI: https://doi.org/10.1016/j.ijbiomac.2018.07.104
Makhado, E.; Pandey, S.; Nomngongo, P. N.; Ramontja, J. J. Colloid Interf. Sci. 2018, 513, 700–714 DOI: https://dx.doi.org/10.1016/j.jcis.2017.11.060 DOI: https://doi.org/10.1016/j.jcis.2017.11.060
Ahmad, R.; Mirza, A. Surf. Interfaces 2017, 8, 65–72 DOI: http://dx.doi.org/10.1016/j.surfin.2017.05.001 DOI: https://doi.org/10.1016/j.surfin.2017.05.001
Jakóbik-Kolon, A.; Bok-Badura, J.; A. K. Milewski, A. K.; Mitko, K. Sep. Sci. Technol. 2018, 53, 1132-1141. DOI: http://dx.doi.org/10.1080/01496395.2017.1298613 DOI: https://doi.org/10.1080/01496395.2017.1298613
Zheng, M.; Lian, F.; Zhu, Y.; Zhang, Y.; Liu, B.; Zhang, L.; Zheng, B. Carbohydr. Polym. 2019, 210, 38–46. DOI: https://doi.org/10.1016/j.carbpol.2019.01.052
Ghorai, S.; Sinhamahpatra, A.; Asish Sarkar A.; Panda, A. B.; Pal, S. Bioresour. Technol. 2012, 119, 181–190. DOI: http://dx.doi.org/10.1016/j.biortech.2012.05.063 DOI: https://doi.org/10.1016/j.biortech.2012.05.063
Peng, X.; Xu, F.; Zhang, W.; Wang, J.; Zeng, C.; Niu, M.; Chmielewská, E. Colloid Surface A. 2014, 443, 27– 36. DOI: http://dx.doi.org/10.1016/j.colsurfa.2013.10.062 DOI: https://doi.org/10.1016/j.colsurfa.2013.10.062
Zhang, S.; Xu, F.; Wang, Y.; Zhang, W.; Peng, X.; Pepe, F. Chem Eng J. 2013, 234, 33–42. DOI: http://dx.doi.org/10.1016/j.cej.2013.08.102 DOI: https://doi.org/10.1016/j.cej.2013.08.102
Koohi, A. D.; Nasimi, F. Chem. Eng. Commun. 2017, 204 (7), 791-802. DOI: http://dx.doi.org/10.1080/00986445.2017.1322071 DOI: https://doi.org/10.1080/00986445.2017.1322071
Ahmad, R.; Mirza, A. Groundwat. Sust. Developm. 2018, 7, 101–108. DOI:https://doi.org/10.1016/j.gsd.2018.03.010
Zhang, W.; Xu, F.; Wang, Y.; Luo, M.; Wang, D. Chem Eng J. 2014, 255, 316–326. DOI:http://dx.doi.org/10.1016/j.cej.2014.06.024 DOI: https://doi.org/10.1016/j.cej.2014.06.024
Perotti, G. F.; Kijchavengkul, T.; Auras, R. A.; Constantino, V.R.L. J. Braz. Chem. Soc. 2017, 28, 649-658 DOI: http://dx.doi.org/10.21577/0103-5053.20160213 DOI: https://doi.org/10.21577/0103-5053.20160213
Patwary, F.; Matsko, N.; Mittal, V. Polym. Compos. 2016, 39, 386-397. DOI: https://dx.doi.org/10.1002/pc.23947 DOI: https://doi.org/10.1002/pc.23947
Gaurav, A.; Ashamol, A.; Deepthi, M. V.; Sailaja, R. R. N. J. Appl. Polym. Sci. 2012, 125, E16–E26 DOI: https://dx.doi.org/10.1002/app.35591 DOI: https://doi.org/10.1002/app.35591
Zimm, B.H.; Bragg, J.K. J. Chem. Phys. 1959, 3, 526-535 DOI: https://dx.doi.org/10.1063/1.1730390 DOI: https://doi.org/10.1063/1.1730390
R Core Team. R: A language and Environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, 2014, http: //www.R-project.org/.
Abbot, L. C.; Batchelor, S. N.; Oakes, J.; Lindsay-Smith, J. R.; Moore, J. N. J. Phys. Chem. B 2004, 108, 13726-13735 DOI: https://doi.org/10.1021/jp0485542
Sato, T.; Norisuye, T.; Fujita, H. Macromolecules 1984, 17, 2696-2700 DOI: https://dx.doi.org/10.1021/ma00142a043 DOI: https://doi.org/10.1021/ma00142a043
Bejanariu, A.; Popa, M.; Le Cerf, D.; Picton, L. Polym. Bull. 2008, 61,631-641 DOI: https://dx.doi.org/10.1007/s00289-008-0987-6 DOI: https://doi.org/10.1007/s00289-008-0987-6
Bejanariu, A.; Popa, M.; Dulong, V.; Picton, L.; Le Cerf, D. Polym. Bull. 2009, 62, 255-538 DOI: https://dx.doi.org/10.1007/s00289-008-0033-8 DOI: https://doi.org/10.1007/s00289-008-0033-8
Rodd, A.B.; Dunstan, D.E.; Boger, D.V.; Schmidt, J.; Burchard, W. Macromol Symp. 2002, 190, 79-91 DOI: https://dx.doi.org/10.1002/masy.200290022 DOI: https://doi.org/10.1002/masy.200290022
Rodd, A. B.; Cooper-White, J.; Dunstan, D. E.; Boger, D. V. Polymer 2001, 42, 185–198 DOI: https://dx.doi.org/10.1016/S0032-3861(00)00311-6 DOI: https://doi.org/10.1016/S0032-3861(00)00311-6
Rodd, A.B.; Dunstan, D.E.; Boger, D.V.; Schmidt, J.; Burchard, W. Macromolecules 2001, 34, 3339-3352 DOI: https://dx.doi.org/10.1021/ma001706g. DOI: https://doi.org/10.1021/ma001706g
Rowsell, J.; Nazar, L.F. J. Am. Chem. Soc. 2000, 122, 3777-3778 DOI: https://dx.doi.org/10.1021/ja993711+ DOI: https://doi.org/10.1021/ja993711+
Chen, Z.; Luan, Z.; Jia, Z.; Li, X. J. Mater. Sci. 2009, 44, 3098–3111 DOI: https://dx.doi.org/10.1007/s10853-009-3412-0 DOI: https://doi.org/10.1007/s10853-009-3412-0
Young, S.L.; Martino, M.; Kienzle-Sterzer, C.; Torres, J. A. J. Sci. Food Agric. 1994, 64, 121-127 DOI: https://doi.org/10.1002/jsfa.2740640118
Oakes, J.; Dixon, S. Rev. Prog. Color 2004, 34, 110-128 DOI: https://doi.org/10.1111/j.1478-4408.2004.tb00156.x
Langmuir, I. J. Am. Chem. Soc. 1916, 38, 2221-2295 DOI: https://doi.org/10.1021/ja02268a002
Freundlich, H. M. F. Z. Phys. Chem. 1906, 57, 385–470 DOI: https://doi.org/ 10.1515/zpch-1907-5723 DOI: https://doi.org/10.1515/zpch-1907-5723
Rafatullah, M.; Sulaiman, O.; Hashim, R.; Ahmad, A. J. Hazard. Mater. 2010, 177, 70-80 DOI: https://doi.org/10.1016/j.jhazmat.2009.12.047
Lozano-Alvarez, J. A.; Jáuregui-Rincón, J.; Mendoza-Díaz, G.; Rodríguez-Vázquez, R.; Frausto-Reyes, C. J. Mex. Chem. Soc. 2009, 53, 59-70.
Hayakawa, K.; Santerre, J. P.; Kwak, J. C. T. Macromolecules 1983, 16, 1642-1645 DOI: https://doi.org/10.1021/ma00244a017
Jain, N.; Trabelsi,S.; Guillot, S.; McLoughlin, D.; Langevin, D.;Letellier, P.; Mireille Turmine, M. Langmuir 2004, 20, 8496-8503 DOI: https://doi.org/10.1021/la0489918
Li, Y. M.; Xu, G. Y.; Chen, A. M.; Yuan, S. L. ; Cao, X. R. J. Phys. Chem. B 2005, 109, 22290-22295 DOI: https://doi.org/10.1021/jp0528414
Lozano-Álvarez, J. A.; Marañón-Ruíz, V. F.; Jáuregui-Rincón, J.; Medina-Ramírez, I.; Frausto-Reyes, C.; Salinas-Gutiérrez, R. J. Mex. Chem. Soc. 2015, 59, 215-227.
Oakes, J.; Gratton, P. J. Chem. Soc., Perkin Trans. 1998, 2, 1857-1864. DOI: https://doi.org/10.1039/A803892I
Sharma, K. K.; O’Neill, P.; Oakes, J.; Batchelor, S. N.; Madhava-Rao, B. S. J. Phys. Chem. A 2003, 107, 7619-7628 DOI: https://doi.org/10.1021/jp035002v
Karukstis, K. K.; Perelman, L. A.; Wong, W. K. Langmuir 2002, 18, 10363-10371 DOI: https://doi.org/10.1021/la020558f
Camesano, T. A.; Wilkinson, K. J. Biomacromolecules 2001, 2, 1184-1191 DOI: https://doi.org/10.1021/bm015555g
Abbott, L. C.; Batchelor, S. N.; Jansen, L.; Oakes, J.; Lindsay-Smith, J. R.; Moore, J. N. New J. Chem. 2004, 28, 815-821 DOI: https://doi.org/10.1039/B401055H
Vahdat, A.; Bahrami, S. H.; Arami, M.; Motahari, A. Radiat. Phys. Chem. 2010, 9, 33–35 DOI: https://doi.org/10.1016/j.radphyschem.2009.08.012
Michniewicz A.; Ledakowicz, S.; Ullrich, R.; Hofrichter, M. Dyes Pigm. 2008, 77, 295-302 DOI: https://doi.org/10.1016/j.dyepig.2007.05.015
Gregory, P., In: The Chemistry and application of dyes, Waring, D. R.; Geoffrey Ha11as, Eds., Plenum Press, New York. EUA, 1990, 17-47 DOI: https://doi.org/10.1007/978-1-4684-7715-3 DOI: https://doi.org/10.1007/978-1-4684-7715-3_2
Silverstein, R.M.; Webster, F. X.; Kiemle, D. J.; Bryce D.L. Spectrometric identification of organic compounds. 8a. ed. John Wiley and Sons, EUA, 2015, 453 pp.
Schrader B. In: Infrared and Raman spectroscopy, methods and applications, B. Schrader, B. Ed., VCH Verlagsgesellschaft mbH, Germany, 1995, 189-222. DOI: https://doi.org/10.1002/9783527615438
Armstrong, D. R.; Clarkson J.; Smith, W. E. J. Phys. Chem. 1995, 99, 17825-17831 DOI: https://doi.org/10.1021/j100051a005
Biswas, N.; Umapathy, S. J. Phys. Chem. A. 2000, 104, 2734-2745 DOI: https://doi.org/10.1021/jp9929263

Downloads
Published
Issue
Section
License
Authors who publish with this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal.








