Synthesis and Catalytic Application of Homoleptic Bis(triazol-5-ylidene)-Silver Complexes
DOI:
https://doi.org/10.29356/jmcs.v70i1.2518Keywords:
Silver, mesoionic carbenes (MICs), catalysis, propargylic aminesAbstract
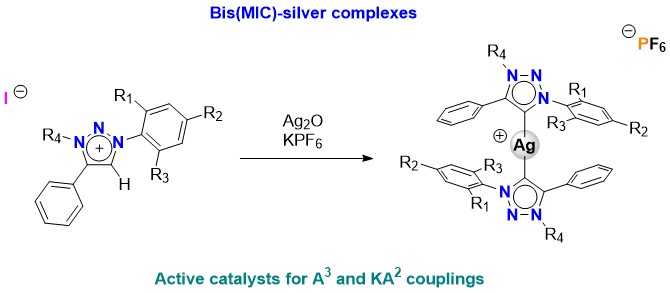
Treatment of 1,2,3-triazolium salts with equimolar amounts of silver oxide (in presence of potassium hexafluorophosphate) generates a series of bis-triazol-5-ylidene silver(I) complexes with the general formula [(MIC)2Ag]PF6 (MIC = triazol-5-ylidene). The new biscarbenic species have been fully characterized including FT-IR and NMR spectroscopies, melting points, and elemental analysis. The catalytic performance of the silver(I) complexes in the solvent free KA2 (ketone-alkyne-amine) coupling for the generation of propargylic amines and the A3 coupling (aldehyde, amine, alkyne) is investigated.
Resumen. El tratamiento de sales de 1,2,3-triazolio con cantidades equimolares de óxido de plata (en presencia de hexafluorofosfato de potasio), genera una serie de complejos de plata soportados por bis(triazol-5-ilidenos) con formula general [(MIC)2Ag]PF6 (MIC = triazol-5-ilideno). Los nuevos complejos de plata(I) han sido completamente caracterizados mediante espectroscopías de FT-IR y RMN, así como puntos de fusión y análisis elementales. El potencial catalítico de los biscarbenos de plata en el acoplamiento KA2 (cetona, aldehído, alquino) para la generación de aminas propargílicas así como el acoplamiento A3 (aldehído, amina, alquino), es investigado.
Downloads
References
1. De Fremont, P.; Scott, N. M.; Stevens, E. D.; Ramnial, T.; Lightbody, O. C.; Macdondald, C. L. B.; Clyburne, J. A. C.; Abernethy, C. D.; Nolan, S. P. Organometallics. 2005, 24, 6301-6309. DOI: https://doi.org/10.1021/om050735i.
2. Wang, H. M. J.; Lin, I. J. B. Organometallics. 1998, 17, 972-975. DOI: https://doi.org/10.1021/om9709704.
3. Guerret, O.; Sole, S.; Gornitzka, H.; Teichert, M.; Trinquier, G.; Bertrand, G. J. Am. Chem. Soc. 1997, 119, 6668-6669. DOI: https://doi.org/10.1021/ja964191a.
4. Tulloch, A. A. D.; Danopoulos, A. A.; Winston, S.; Kleinhenz, S.; Eastham, G. Dalton Trans. 2000, 24, 4499-4506. DOI: https://doi.org/10.1039/B007504N.
5. Caballero, A.; Diez-Barra, E.; Jalon, F. A.; Merino, S.; Tejeda, J. J. Organomet. Chem., 2001, 617-618, 395-398. DOI: https://doi.org/10.1016/S0022-328X(00)00715-4.
6. Maishal, T. K.; Basset, J.-M.; Boualleg, M.; Coperet, C.; Veyre, L.; Thieuleux, C. Dalton Trans. 2009, 35, 6956-6959. DOI: https://doi.org/10.1039/B900136K.
7. Lin, I. J. B.; Vasam, C. S. Coord. Chem. Rev. 2007, 251, 642-670. DOI: https://doi.org/10.1016/j.ccr.2006.09.004.
8. Lin, I. J. B.; Vasam, C. S. Comm. Inorg. Chem. 2004, 25, 75-129. DOI: https://doi.org/10.1080/02603590490883652
9. Ceramella, J.; Catalano, A.; Mariconda, A.; D´Amato, A.; Aquila, S.; Saturnino, C.; Rosano, C.; Sinicropi, M. S.; Longo, P. Pharmaceuticals. 2025, 18, 9. DOI: https://doi.org/10.3390/ph18010009.
10. T. Xu, X. Mu, H. Peng, G. Liu, Angew. Chem. Int. Ed. 2011, 50, 8176–8179. DOI: https://doi.org/10.1002/anie.201103225.
11. S. Sakai, A. Fujioka, K. Imai, K. Uchiyama, Y. Shimizu, K. Higashida, M. Sawamura, Adv. Synth. Catal. 2022, 364, 2333–2339. DOI: https://doi.org/10.1002/adsc.202200327.
12. Cervantes-Reyes, A.; Saxl, T.; Stein, P. M.; Rudolph, M.; Rominger, F.; Asiri, A. M.; Hashmi, A. S. K. ChemSusChem. 2021, 14, 2367-2374. DOI: https://doi.org/10.1002/adsc.202200327.
13. Gründemann, S.; Kovacevic, A.; Albrecht, M.; Faller, J.; Crabtree, R. H. Chem. Commun. 2001, 21, 2274-2275. DOI: https://doi.org/10.1039/B107881J.
14. Vivancos, A.; Segarra, C.; Albrecht, M. Chem. Rev. 2018, 118, 9493-9586. DOI: https://doi.org/10.1021/acs.chemrev.8b00148.
15. Patricio-Rangel, E. B.; Salazar-Pereda, V.; Cortezano-Arellano, O.; Mendoza-Espinosa, D. Dalton Trans. 2022, 51, 2641-2651. DOI: https://doi.org/10.1039/D1DT04221A.
16. Kolb, H. C.; Finn, M. G.; Sharpless, K. B. Angew. Chem. Int. Ed. 2001, 40, 2004-2021. DOI: https://doi.org/10.1002/1521-3773(20010601)40:11<2004::AID-ANIE2004>3.0.CO;2-5.
17. Hettmanczyk, L.; Schulze, D.; Suntrup, L.; Sarkar, B. Organometallics. 2016, 35, 3828–3836. DOI: https://doi.org/10.1021/acs.organomet.6b00675.
18. Poulain, A.; Canseco-Gonzalez, D.; Hynes-Roche, R.; Müller-Bunz, H.; Schuster, O.; Stoeckli-Evans, H.; Neels, A.; Albrecht, M. Organometallics. 2011, 30, 1021–1029. DOI: https://doi.org/10.1021/om101076u.
19. Bouffard, J.; Keitz, B. K.; Tonner, R.; Guisado-Barrios, G.; Frenking, G.; Grubbs, R. H.; Bertrand, G. Organometallics. 2011, 30, 2617–2627. DOI: https://doi.org/10.1021/om200272m.
20. Beillard, A.; Bantreil, X.; Metro, T.-X.; Martinez, J.; Lamaty, F. Dalton Trans. 2016, 45, 17859-17866. DOI: https://doi.org/10.1039/C6DT03564G.
21. Vessally, E.; Hosseinian, A.; Edjlali, L.; Bekhradnia, A.; Esrafili, M. D. RSC Adv. 2016, 6, 71662-71675. DOI: https://doi.org/10.1039/C6RA08720E.
22. Jesin, I.; Nandi, G. C. Eur. J. Org. Chem. 2019, 2019, 2704-2720. DOI: https://doi.org/10.1002/ejoc.201900001.
23. Abbiat, G.; Rossi, E.; Beilstein J. Org. Chem. 2014, 10, 481-513. DOI: https://doi.org/10.3762/bjoc.10.46.
24. Wei, C.; Li, Z.; Li, C.-J. Org. Lett. 2003, 5, 4473-4475. DOI: https://doi.org/10.1021/ol035781y
25. D´Amato, A.; Sirignano, M.; Russo, S.; Troiano, R.; Mariconda, A.; Longo, P. Catalysts. 2023, 13, 81. DOI: https://doi.org/10.3390/catal13050811.
26. Mariconda, A.; Sirignano, M.; Costabile, C.; Longo, P. Mol. Catal. 2020, 480, 110570. DOI: DOI: https://doi.org/10.1016/j.mcat.2019.110570.
27. Beillar, A.; Metro, T.-X.; Bantreil, X.; Martinez, J.; Lamaty, F. Eur. J. Org. Chem. 2017, 2017, 4642. DOI: https://doi.org/10.1002/ejoc.201700985.
28. Mateus, M.; Kiss, A.; Cisarova, I.; Karpinski, T. M.; Rycek, L. Appl. Organomet. Chem. 2023, 37, e6994. DOI: https://doi.org/10.1002/aoc.6994.
29. Mateus, M.; Rycek, L. ChemPlusChem. 2024, 89, e202400365. DOI: https://doi.org/10.1002/cplu.202400365.
30. Ruiz-Mendoza, F. J.; Campos-Dominguez, E.; Torres-Salazar, M. G.; Alvarez-Hernandez, A.; Mendoza-Espinosa, D., ChemPlusChem. 2025, 90, e202400765. DOI: https://doi.org/10.1002/cplu.202400765.

Downloads
Published
Issue
Section
License
Copyright (c) 2026 Hugo Omar Quezada-Márquez, Emmanuel Campos-Dominguez, Leonardo Kaleb Méndez-Jiménez, Daniel Mendoza-Espinosa

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Authors who publish with this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal.








