Journal of the Mexican Chemical Society, vol. 64, núm. 3, 2020
Sociedad Química de México A.C.
Oscar Abelardo Ramírez-Marroquín *
Universidad del Papaloapan, Mexico
María Adelina Jiménez-Arellanes
Instituto Mexicano del Seguro Social, Mexico
Julieta Luna-Herrera
Instituto Politécnico Nacional, Mexico
José Luis Olivares-Romero
Instituto de Ecología, Mexico
Israel Bonilla-Landa
Instituto de Ecología, Mexico
Karla Viridiana Castro-Cerritos
Universidad del Papaloapan, Mexico
Recibido: 07 Febrero 2020
Aceptado: 24 Abril 2020
Resumen: En la presente investigación se evaluó la actividad anti-inflamatoria in vivo de seis amidas α,β-insaturadas, identificadas en la literatura como piperlotinas debido a su inicial aislamiento a partir de especies vegetales del género Piper (como las pimientas). La reacción del β-amidofosfonato 4 con un aldehído aromático y K2CO3 en molienda con mortero y pistilo produjo las piperlotinas 5-6 con buenos rendimientos (70-80%) en tiempos cortos de reacción, obteniendo solamente el diastereoisómero (E). Las piperlotinas preparadas se evaluaron como agentes anti-inflamatorios y antibacterianos, observando excelente actividad anti-inflamatoria para los derivados 2 y 6, especialmente cuando se probaron mediante administración tópica (modelo de inflamación aguda por TPA). Además, la piperlotina A y los derivados 2 y 6 mostraron actividad antibacteriana contra Mycobacterium tuberculosis (MIC = 50 µg/mL). En conclusión, la síntesis de piperlotinas a través de molienda en condiciones libres de disolvente generó importantes productos que pueden ser utilizados como punto de partida para generar nuevos compuestos bioactivos para el tratamiento de padecimientos relacionados con la inflamación.
Palabras clave: Mecanoquímica, piper amidas, inflamación, tuberculosis.
Abstract: In the present study we evaluated six α,β-unsaturated amides named piperlotines (for being isolated originally from Piper species) as new in vivo anti-inflammatory agents. In addition, we report the mechanosynthesis of two of them by mechanochemical activation of a Horner-Wadsworth-Emmons reaction. The reaction of β-amidophosphonate 4, an aromatic aldehyde and K2CO3 under grinding in a mortar and pestle afforded piperlotines 5-6 in good yields (70-88%) in short reaction times, obtaining only (E)-diastereomer. Piperlotines previously prepared were tested as anti-inflammatory and antibacterial agents. In this respect, derivatives 2 and 6 exhibited excellent in vivo anti-inflammatory activity on mice, especially trough topical administration (TPA acute inflammation model). Furthermore, piperlotine A, and compounds 2 and 6 had slight antimycobacterial activity against Mycobacterium tuberculosis (MIC = 50 µg/mL). In conclusion, the solvent-free mechanosynthesis of piperlotines produced valuable compounds that could serve as templates for further investigation in the search of better drug-like compounds for the treatment of inflammatory diseases.
Keywords: Mechanochemistry, piper amides, inflammation, tuberculosis.
Introduction
Piperlotines are very important secondary metabolites which have been isolated from different Piper species (Piperaceae) [1]; such valuable compounds have been just partially explored in search of interesting pharmacological activities, for example as antiplatelet aggregation [2] and anticonvulsant agents [3-4].
Nonetheless, pharmacological applications of these α,β-unsaturated amides and related derivatives still remain underexplored. For example, inflammation-related diseases as arthritis, psoriasis, lupus, allergy and asthma, are important public health problems all over the world [5-7] but actually, to the best of our knowledge no one in vivo inflammation test has been undertaken using piperlotines as potential anti-inflammatory agents.
In other instance, tuberculosis (TB) is a health problem especially in developing countries, being one of the top ten causes of death by infectious processes and affecting a quarter of the world population, in addition to the increase of multidrug-resistant and extensively drug-resistant cases [8] that motivate the search of new anti-mycobacterial therapies. In this respect, α,β-unsaturated amides, including amide derivatives of cinnamic acid, have been proposed as alternative or complementary anti-tubercular agents in multiple studies [9-12].
Few methods have been reported for the synthesis of piperlotines and derivatives, including: alkyne’s catalyzed aminocarbonylation [13], transamidation from another unsaturated amides [14], amidation of cinnamic acids [15-17] and olefination of aromatic aldehydes by means of the Horner-Wadsworth-Emmons reaction [18].
Mechanosynthesis is a solvent-free method used to generate a wide variety of compounds, including molecules of pharmacological interest [19, 20], in this regard, our group has developed a simple, fast, and diastereoselective method to obtain piperlotines A, C and their derivatives through the mechanical activation of a β-amidophosphonate intermediate [21].
In this work, piperidine-derived α,β-unsaturated amides 5, 6 were prepared by means of mechanochemical activation of a Horner-Wadsworth-Emmons reaction (HWE) between a β-amidophosphonate 4 and an aromatic aldehyde in alkaline media under solvent-free conditions.
Then, compounds 5,6 and previously synthesized ones were then tested as anti-inflammatory agents through two in vivo acute inflammation models (Carrageenan, by oral administration and TPA through topical administration). In addition, their potential anti-tubercular activity was tested on Mycobacterium tuberculosis (MTb) H37Rv strain (ATCC 27294).
Experimental
Materials and methods
All commercial materials were used as received from Sigma-Aldrich® without further purification. Flash chromatography was performed using Merck 230-400-mesh Silica Flash 60® silica gel. Thin-layer chromatography was performed with pre-coated TLC sheets of silica gel (60F254, Merck). NMR spectra were recorded in a Bruker Ascend instrument (500 MHz for 1H, 126 MHz for 13C, and 202 MHz for 31P, 470 MHz for 19F), and calibrated with CDCl3 as the solvent and TMS as the internal standard signal. Chemical shifts (δ) are reported in parts per million. Multiplicities are recorded as follows: s = singlet, d = doublet, t = triplet, dd = doublet of doublets, td = triplet of doublets, bs = broad singlet, q = quartet, and m = multiplet. Coupling constants (J) are given in Hz. High-resolution mass spectra (HRMS) were obtained in a Synapt G2-Si (Waters) spectrometer equipped with electrospray ion source (ESI), single quadrupole mass filter and time of flight mass analyzer (Q-TOF). Mass spectrometer was operated in positive mode, calibrated with NaI and exact mass was corrected with leucine enkephaline (556.2771).
Synthesis of N-(bromoacetyl)piperidine 3
To an ice bath-cooled solution of bromoacetyl bromide (12.65 mmol) in 20 mL of CH2Cl2 was added dropwise via addition funnel a solution of piperidine (25.31 mmol) in 30 mL of CH2Cl2, and the reaction mixture was stirred for 20 h at room temperature. Then the reaction mixture was quenched by the addition of 25 mL of distilled water and extracted with CH2Cl2 (3 × 25 mL). Next, to the combined organic extracts 15 mL of HCl 0.2 N was added. Then, organic partition was washed with distilled water. Finally, the organic extract was dried over Na2SO4, filtered, and evaporated under reduced pressure, yielding 2.226 g (86%) of a pale-yellow oil. 1H and 13C NMR spectra were found to be identical with the ones described in [22].
Synthesis of diethyl [2-oxo-2-(piperidin-1-yl)ethyl]phosphonate 4
Triethyl phosphite (2.16 mmol) was added to alkyl bromide 3 (1.97 mmol) at room temperature. Next, reaction mixture was stirred at 60 °C for 8 h. The crude reaction was purified through flash column chromatography (EtOAc:MeOH 95:05), affording 0.433 g (84%) of a pale-yellow oil. 1H NMR (500 MHz, CDCl3): δ = 1.34 (t, J = 7.1 Hz,6H, CH3), 1.54-1.64 (m, 6H, CH2), 3.07 (d, J = 22.0 Hz, 2H, CH2P), 3.49-3.58 (m, 4H, CH2N), 4.14-4.20 (m, 4H, CH2O) ppm; 13C NMR (125 MHz, CDCl3): δ = 16.4 (d, J = 6.3 Hz, CH3), 24.3 (CH2), 25.5 (CH2), 26.3 (CH2), 33.4 (d, J = 133.5 Hz, CH2P), 43.1 (CH2N), 48.2 (CH2N), 62.6 (d, J = 6.5 Hz, CH2O), 163.0 (d, J = 5.6 Hz, C=O) ppm; 31P NMR (202 MHz, CDCl3): δ = 21.6 ppm; ESI(+)-HRMS : m/z 264.1372 (calculated 264.1360).
General procedure for the synthesis of piperlotines A, C and derivatives 1-2, 5-6
1 equivalent of phosphonate 4, 1.5 equivalents of K2CO3 and 1.1 equivalents of the corresponding aldehyde (4-nitrobenzaldehyde and 4-trifluoromethylbenzaldehyde) were grinded with a mortar and pestle for 30 min under solvent-free conditions. Afterwards, the crude reaction mixture was purified through flash column chromatography (EtOAc:Hexane 50:50), affording desired products. Full method of synthesis and experimental characterization of Piperlotines A, C, 1-2 could be consulted in ref. 21.
(E)-3-(4-nitrophenyl)-1-(piperidin-1-yl)prop-2-en-1-one (5) Yield: 135 mg (88%) as a white solid; m.p.: 173-174.5 °C; 1H NMR (500 MHz, CDCl3): δ = 1.64-1.66 (m, 4H, CH2-CH2), 1.70-1.74 (m, 2H, CH2-CH2), 3.60-3.62 (m, 2H, CH2-N), 3.68-3.70 (m, 2H, CH2-N), 7.06 (d, J = 15.5 Hz, 1H, CH), 7.65-7.68 (m, 3H, CH), 8.24 (AA’ BB’, J = 8.5 Hz, 2H, Harom) ppm; 13C NMR (126 MHz, CDCl3): δ = 24.56 (CH2-CH2), 25.58 (CH2-CH2), 26.82 (CH2-CH2), 43.48 (CH2-N), 47.16 (CH2-N), 122.15, 124.10, 128.25, 139.36, 141.76, 147.93, 164.28 (C=O) ppm; ESI(+)-HRMS: m/z 261.1241 (calculated 261.1234).
(E)-1-(piperidin-1-yl)-3-[4-(trifluoromethyl)phenyl]prop-2-en-1-one (6) Yield: 132 mg (70 %) as a white solid; m.p.: 87-88 °C; 1H NMR (500 MHz, CDCl3): δ = 1.64-1.71 (m, 6H, CH2-CH2), 3.60-3.70 (m, CH2-N), 7.0 (d, J = 15.5 Hz, 1H, CH=CH), 7.62 (m, 4H, Harom) 7.65 (d, J = 15.5 Hz, 1H, CH=CH) ppm; 13C NMR (126 MHz, CDCl3): δ = 24.6 (CH2-CH2), 25.6 (CH2-CH2), 26.8 (CH2-CH2), 43.4 (CH2-N), 47.1 (CH2-N), 124.0 (q, J = 272.1 Hz, CF3), 125.7 (q, J = 3.7 Hz), 130.9 (q, J = 32.6 Hz, Carom), 138.9, 140.4, 164.7 (C=O) ppm; 19F (470 MHz, CDCl3): δ = -62.7 ppm; ESI(+)-HRMS: m/z 284.1280 (calculated 284.1257).
Pharmacological assays
Antimycobacterial activity
The antimycobacterial activity was carried out by means of the microplate alamar blue assay (MABA), a well stablished method for determining susceptibility of Mycobacterium tuberculosis to medicinal plant extracts and pure compounds [23-25]. In this work, M. tuberculosis H37Rv [ATCC 27294], strain sensitive to streptomycin (STR), isoniazid (IHN), rifampicin (RIF), ethambutol (EMB), or pyrazinamide (PZA)] was used according to a previously described method [26]. Each compound (10 mg) was solubilized in 500 µL of DMSO and from these stock solutions, several dilutions were prepared to achieve concentrations between 200 and 3.13 µg/mL. The maximum DMSO concentration used in the MABA assay does not affect mycobacterial growth.
Mycobacteria were grown for 7 days at 37°C in a Middlebrook 7H9 medium supplemented with 0.2% glycerol and 10% DCOA (dextrose, catalase, oleic acid, and albumin). The mycobacterial suspension was adjusted in a no. 1 tube of a McFarland nephelometer and diluted 1:50 with the same medium to obtain 6 × 106 Colony Forming Units (CFU)/mL. An assay was performed on sterile 96-wells microplate and two controls (microorganism and solvent) were included for H37Rv strains.
Carrageenan acute inflammation model
Carrageenan-induced paw edema procedure was conducted on male BALB/c mice as previously described [26]. It is important to note than carrageenan model is a broadly used method for determining oral anti-inflammatory activity of medicinal plant extracts, pure compounds and biological products [27-29]. Briefly, treated groups (n=5) received indomethacin (20 mg/kg, drug reference) or tested compounds (25 mg/kg) by the intragastric route 1 h prior to the injection of carrageenan (20 µL, 2%). The samples were solubilized in 10 % Tween 80 and the control received only vehicle. The percentage of inhibition was calculated by comparing the measurement of the paw edema at different times (1, 3, 5, 7, and 24 h) using a digital micrometer and the value of time zero (baseline) (E0). The results were analyzed with the formula:
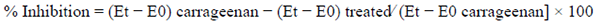
12-O-tetradecanoylphorbol-13-acetate (TPA) acute inflammation model
TPA-induced ear edema was conducted on male BALB/c mice as previously described [26] and it has been extensively used for determining topical anti-inflammatory activity of medicinal plant extracts and pure compounds [30-32]. Control was treated with TPA (2.5 μg) in acetone on the right ear (W’s) and then left ear received only 25 µL of acetone (Wo). The experimental groups (n=5) received TPA and 30 min later were treated with test compounds (2 mg/ear) in the right ear (Ws). Anti-inflammatory activity was calculated according to the weight difference between the ear sections (6 mm) at 6 h, compared with the control group, using the following formula:
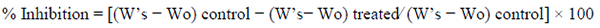
Results and discussion
Rational drug design is a modern strategy followed to discover new bioactive compounds and for to develop new drugs. In this regard, computer aided drug design (CADD) is a powerful tool for prediction of potential pharmacological activity. For example, the Prediction of Activity Spectra for Substances (PASS Online) [33] database is a freely available resource successfully utilized for prediction of pharmacological activity of known or novel organic compounds. With this on mind, and according to our interest in the synthesis of Piper amides we envisaged the synthesis and pharmacological evaluation of piperlotines structurally related to anti-inflammatory natural products and commercial drugs as piperine [34-36], idrocilamide [37], tranilast [38], and E93 [39] (Fig. 1) which contains: an α,β-unsaturated amide conjugated with one or more C=C double bonds and an aromatic ring. Alkoxy substituents are very common in the aromatic portion of Piper amides’ molecular structure, including the above-named anti-inflammatory compounds. In this regard, we considered to include electron-donating (R = OCH3, EDG) substituents into aromatic ring of Piperlotine A, Piperlotine C and 1. Additionally, an electron-withdrawing (R = NO2, CF3, EWG) substituent was introduced in 5,6 in order to determine the effect of electronic density on pharmacological activity (Fig. 1).
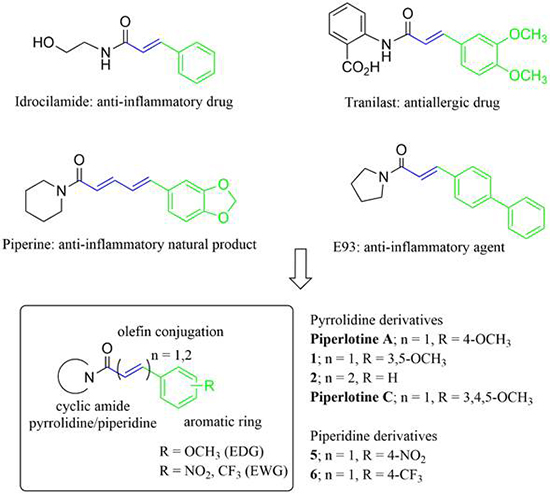
Fig. 1
Anti-inflammatory α,β-unsaturated amides structurally related to proposed piperlotines.
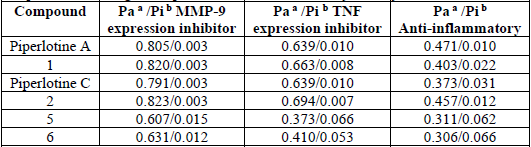
Next, piperlotines A, C and derivatives were predicted as potential anti-inflammatory agents (probability of activity from 0.31 to 0.47, being 1 the maximal probability of activity) through PASS prediction (Table 1). Altogether, above named derivatives obtained interesting prediction as matrix-metalloproteinase-9 (MMP-9) expression inhibitors (0.61-0.82) (Table 1). Now, it is well established a relationship between increased levels of matrix-metalloproteinases and the development of acute or chronic inflammation processes in diverse tissues [40]. Moreover, inflammatory-related diseases as arthritis and systemic lupus erythematosus have been correlated with unusual tissular matrix-metalloproteinases concentration [41-42]. In addition, target amides were predicted as tumor necrosis factor (TNF) expression inhibitors (Pa from 0.373 to 0.694), another important pro-inflammatory mediator [43].
Finally, all compounds agreed with Lipinski’s rules as drug-like criteria [44]. In view of all above mentioned, piperlotine-like amides were ideal candidates for pharmacological tests as anti-inflammatory agents.
PASS prediction of target compounds on anti-inflammatory activity.
Consequently, the reaction of piperidine and bromoacetyl bromide produced amide 3 in 86% yield, which when treated with triethyl phosphite afforded β-amidophosphonate 4 in 84% yield. Finally, 4 was reacted with an aromatic aldehyde and K2CO3 in a mortar and pestle under solvent-free conditions [21], affording products (E)-5, 6 in 70-88% yield in just 30 min (Scheme 1).
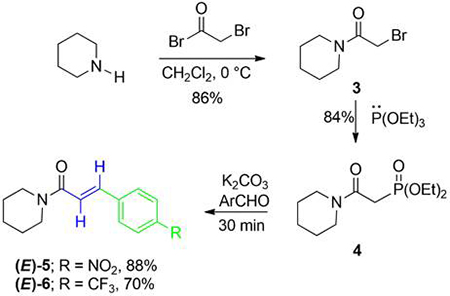
Scheme 1
Synthesis of 5-6.
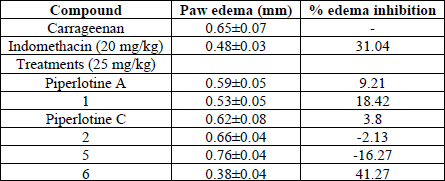
Next, synthesized compounds 5-6 and previously prepared piperlotines A, C and 1-2 were tested through carrageenan induced acute inflammation model. Structure-activity relationships (SAR) studies indicated the aromatic substitution pattern in tested derivatives was a strong determinant of anti-inflammatory activity. In this regard, presence of EDG (R = OCH3) in phenyl ring of piperlotines A, C and compound 1 determined its low edema inhibition, compared with positive control -indomethacin- (Table 2). The extension of olefin conjugation in 2 had a markedly deleterious effect in its anti-inflammatory activity. Furthermore, the presence of an EWG (R = CF3) in phenyl ring greatly favored edema inhibition in 6 (having most potent anti-inflammatory activity than indomethacin, being the most active). Notwithstanding, the presence of nitro group in 5 produced a pronounced pro-inflammatory effect. It is worth to mentioning that nitro and trifluoromethyl groups exert its electron-withdrawing effect through different mechanisms; nitro group by resonance effect, but trifluoromethyl group by inductive effect. Additionally, the introduction of trifluoromethyl substituent has proven to improve absorption and metabolic stability of many bioactive compounds [45-46].
Anti-inflammatory activity of tested compounds on carrageenan model after 5 h.
Afterwards, encouraging results were observed when synthesized compounds were tested on TPA acute inflammation model. This time, all compounds were anti-inflammatory in a range of 31.68-52.02 % edema inhibition. Once again trifluoromethyl derivative 6 was the more potent hit, almost comparable to indomethacin (60.32%) (Table 3). Identical edema inhibition was observed for derivative 2 (52.02%), being both the more lipophilic compounds of the series (logP = 3.82, 2.94, respectively) (Table 3), and thereby having a better absorption through mice skin. Afterwards, it was observed that the more methoxy groups were present in the aromatic ring, the more potency was achieved. Thus, edema inhibition of piperlotine C > 1 > piperlotine A (42.24%, 41.76% and 32.21%, respectively) (Table 3). Finally, the nitro derivative 5 was the less active hit of the series (31.68% edema inhibition). Nonetheless, it is noticeable than 5 exhibited anti-inflammatory activity by TPA topical model, but pro-inflammatory activity was observed when carrageenan model was carried out. Additional experiments are needed to clarify this dual behavior previously observed in our group [47].
Anti-inflammatory activity of tested compounds on TPA model.
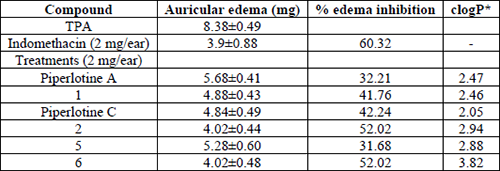
In conclusion, the topical anti-inflammatory activity of 2 and 6 was remarkably in comparison to positive control -indomethacin-. More interesting, compound 6, the more active hit trough oral and topical administration has been previously described as an anticonvulsant agent on mice [3-4] and it could serve as a useful scaffold in the search of potent central nervous system anti-inflammatory agents.
On the other hand, α,β-unsaturated amides have been probed as antimycobacterial agents in several works [9-12, 49], especially piperine amide analogs which had more potent antimycobacterial activity than first line drugs as ethambutol and isoniazid on Mycobacterium tuberculosis H37Rv strain [50]. Based on this, we tested synthesized piperlotines against MTb, where only piperlotine A and compounds 2 and 6 showed slight activity (MIC = 50 µg/mL) (Table 4) compared to isoniazid and rifampicin (MIC < 1 µg/mL) [11-12], but comparable to other structurally related Piper α,β-unsaturated amides as: sarmentine, brachyamide B and guineensine (MIC = 50 µg/mL) [51] and higher than cinnamic acid itself (MIC = 100 µg/mL) [11].
Antibacterial activity of tested compounds on MTb H37Rv.
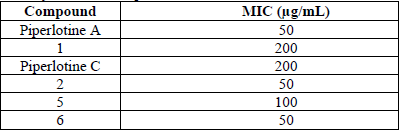
Furthermore, several authors have reported synergistic activity of cinnamic acid amide derivatives [12, 52-53] in combination with isoniazid or rifampicin. Then, we suggest piperlotines synthesized in this work could be used in combination with classical treatments on MTb strains.
Conclusion
Two piperlotine-like compounds 5-6 were easily synthesized from β-amido phosphonate 4 and an aromatic aldehyde in good yields. No solvent or additional heating was needed and only (E)-diastereomer was obtained. Next, synthesized and previously obtained piperlotines were tested as in vivo anti-inflammatory agents for the first time. In this respect, trifluoromethyl derivative 6 had remarkably anti-inflammatory activity in two acute inflammation models on mice (comparable or higher than indomethacin, the commercial anti-inflammatory drug reference), being active through oral and topical administration. Finally, piperlotine A, and derivatives 2, 6 exhibited slight antibacterial activity on MTb H37Rv strain, and could be used in combination with first line antituberculosis drugs in future investigations, pointing the potential of piperlotines and its derivatives to further medicinal chemistry research.
References
1. Gómez‐Calvario, V.; Rios, M.Y. Magn. Reson. Chem. 2019, 57, 1-77 DOI: https://doi.org/10.1002/mrc.4857
2. Li C-Y.; Tsai, W-J.; Damu, A. G.; Lee, E-J.; Wu, T-S.; Dung, N. X.; Thang, T. D.; Thanh, L. J. Agric. Food Chem. 2007, 55, 9436-9442 DOI: https://doi.org/10.1021/jf071963l
3. Hou, T.; Liao, N.; Xu, X.; Li, Y.J. Mol. Mod.2000, 6, 438-445 DOI: ://doi.org/10.1007/s0089400060438
4. Hou, T. J.; Wang, J. M.; Liao, N.; Xu, X. J.J. Chem. Inf. Comput. Sci.1999,39, 775-781 DOI: https://doi.org/10.1021/ci990010n
5. Alinaghi, F.; Calov, M.; Kristensen, L. E.; Gladman, D. D.; Coates, L. C.; Jullien, D.; Gottlieb, A. B.; Gisondi, P.; Wu, J. J.; Thyssen, J. P.; Egeberg, A. J. Am. Acad. Dermatol. 2019, 80, 251-265 DOI: https://doi.org/10.1016/j.jaad.2018.06.027
6. Barbour, K. E.; Helmick, C. G.; Boring, M.; Brady, T. J. MMWR Morb. Mortal Wkly. Rep. 2017, 66, 246-253 DOI: http://dx.doi.org/10.15585/mmwr.mm6609e1
7. The Global Asthma Report 2018. Auckland, New Zealand: Global Asthma Network, 2018.
8. World Health Organization (2019) Global tuberculosis report 2019. Geneva: 2019. Licence: CC BY-NC-SA 3.0 IGO.
9. Kakwani, M. D.; Suryavanshi, P.; Ray, M.; Rajan, M.; Majee, S.; Samad, A.; Devarajan, P.; Degani, M. S., Bioorganic & medicinal chemistry letters 2011, 21, 1997-1999 DOI: 10.1016/j.bmcl.2011.08.076
10. Wu, Z.-R.; Zhi, D.-J.; Zheng, L.-F.; Li, J.-Y.; Li, Y.; Xie, Q.-J.; Feng, N.; Bao, Y.-F.; Gao, Q.-Y.; Song, Y., Medicinal Chemistry Research 2015, 24, 161-170 DOI: 10.1007/s00044-014-1112-z
11. Rastogi, N.; Goh, K. S.; Horgen, L.; Barrow, W. W., FEMS Immunology & Medical Microbiology 1998, 21, 149-157. DOI: 10.1111/j.1574-695X.1998.tb01161.x
12. Carvalho, S. A.; da Silva, E. F.; de Souza, M. V.; Lourenço, M. C.; Vicente, F. R., Bioorganic & medicinal chemistry letters 2008, 18, 538-541 DOI: 10.1016/j.bmcl.2007.11.091
13. Driller, K. M.; Prateeptongkum, S.; Jackstell, R.; Beller, M. Angew. Chem. Int. Ed. Engl. 2011, 50, 537-541 DOI: 10.1002/anie.201005823
14. Gowri, P. M.; Radhakrishnan, S. V. S.; Rao, V. R. S.; Madnu, A.; Hymavathi, A.; Rao, J. M. Curr. Sci. 2015, 109, 1775-1777
15. Tavares-da-Silva, E. J.; Varela, C. L.; Pires, A. S.; Encarnação, J. C.; Abrantes, A. M.; Botelho, M. F.; Carvalho, R. A.; Proença, C.; Freitas, M.; Fernandes, E.; Roleira, F. M. F. Bioorg. Med. Chem. 2016, 24, 3556-3564 DOI: 10.1016/j.bmc.2016.05.065
16. da Silva-Carrara, V.; Ferreira-Cunha-Júnior, E.; Torres-Santos, E. C.; Gonçalves-Correa, A.; Monteiro, J. L.; Galhardo-Demarchi, I.; Campana-Lonardoni, M. V.; Garcia-Cortez, D. A. Braz. J. Pharmacog. 2013, 23, 447-454. DOI: 10.1590/S0102-695X2013005000022
17. Guan, L.-P.; Wei, C.-X.; Deng, X.-Q.; Sui, X.; Piao, H.-R.; Quan, Z.-S. Eur. J. Med. Chem. 2009, 44, 3654-3657 DOI: 10.1016/j.ejmech.2009.02.015
18. Kim, S.; Lim, C.; Lee, S.; Lee, S.; Cho, H.; Lee, J.-Y.; Shim, D.-S.; Park, H. D.; Kim, S. ACS Comb. Sci. 2013, 15, 208-215 DOI: 10.1021/co400003d
19. Metro, T. X.; Bonnamour, J.; Reidon, T.; Sarpoulet, J.; Martinez, J.; Lamaty, F., Chemical communications 2012, 48, 11781-3 DOI: 10.1039/c2cc36352f
20. Tan, D.; Strukil, V.; Mottillo, C.; Friscic, T., Chemical communications 2014, 50, 5248-50 DOI: https://doi.org/10.1039/c3cc47905f
21. Ramírez-Marroquín, O. A.; Manzano-Pérez, F.; López-Torres, A.; Hernández-López, A.; Cortés-Pacheco, A.; Reyes-González, M. A. Synth. Commun. 2019, 49, 244-255 DOI: 10.1080/00397911.2018.1550204
22. Leung, P. S-W.; Teng, Y.; Toy, P. H. Org. Lett. 2010, 12, 4996-4999 DOI: 10.1021/ol1021614
23. Nguta, J. M.; Appiah-Opong, R.; Nyarko, A. K.; Yeboah-Manu, D.; Addo, P. G. A.; Otchere, I.; Kissi-Twum, A. J. Ethnopharmacol. 2016, 182, 10-15 DOI: 10.1016/j.jep.2016.02.010.
24. Dzul-Beh, A. J.; García-Sosa, K.; Uc-Cachón, A. H.; Bórquez, J.; Loyola, L. A.; Barrios-García, H. B.; Peña-Rodríguez, L. M.; Molina-Salinas, G. M. Braz. J. Pharmacog. 2019, 29, 798-800 DOI: 10.1016/j.bjp.2019.06.001
25. Doğan, H.; Doğan, S. D.; Gündüz, M. G.; Krishna, V. S.; Lherbet, C.; Sriram, D.; Sahin, O.; Saripinar, E. Eur. J. Med. Chem. 2020, 188, 112035 DOI: 10.1016/j.ejmech.2020.112035
26. Pérez-González, M. Z.; Gutiérrez-Rebolledo, G. A.; Yépez-Mulia, L.; Rojas-Tomé I. S.; Luna-Herrera, J.; Jiménez-Arellanes, M. A. Biomed. Pharmacother. 2017, 89, 89-97 DOI: 10.1016/j.biopha.2017.02.021
27. Solanki, H. K.; Shah, D. A.; Maheriya, P. M.; Patel, C. A. Int. J. Biol. Macromol. 2015, 72, 1277-1282 DOI: 10.1016/j.ijbiomac.2014.09.059
28. Jisha, N.; Vysakh, A.; Vijeesh, V.; Latha, M. S. Pathophysiology 2019, 26, 323-330 DOI: 10.1016/j.pathophys.2019.08.002
29. Xu, X-T.; Mou, X-Q.; Xi, Q-M.; Liu, W-T.; Liu, W-F.; Sheng, Z-J.; Zheng, X.; Zhang, K.; Du, Z-Y.; Zhao, S-Q.; Wang, S-H. Bioorg. Med. Chem. Lett. 2016, 26, 5334-5339 DOI: 10.1016/j.bmcl.2016.09.034
30. Xu, X-T.; Mou, X-Q.; Xi, Q-M.; Liu, W-T.; Liu, W-F.; Sheng, Z-J.; Zheng, X.; Zhang, K.; Du, Z-Y.; Zhao, S-Q.; Wang, S-H. Bioorg. Med. Chem. Lett. 2016, 26, 5334-5339 DOI: 10.1016/j.bmcl.2016.09.034
31. Figueroa-Suárez, M. Z.; González-Christen, J.; Cardoso-Taketa, A. T.; Gutiérrez-Villafuerte, M. C.; Rodríguez-López, V. J. Ethnopharmacol. 2019, 238, 111786 DOI: 10.1016/j.jep.2019.03.013
32. Madrigal, D. A.; Escalante, C. H.; Gutiérrez-Rebolledo, G. A.; Cristobal-Luna, J. M.; Gómez-García, O.; Hernández-Benitez, R. I.; Esquivel-Campos, A. L.; Pérez-Gutiérrez, S.; Chamorro-Cevallos, G. A.; Delgado, F.; Tamariz, J. Bioorg. Med. Chem. 2019, 27, 115053 DOI: 10.1016/j.bmc.2019.115053
33. Druzhilovskiy, D.; Rudik, A.; Filimonov, D.; Lagunin, A.; Gloriozova, T.; Poroikov, V. Russ. Chem. Bull. 2016, 65, 384-393 DOI: 10.1007/s11172-016-1310-6
34. Bang, J. S.; Oh, D. H.; Choi, H. M.; Sur, B-J.; Lim, S-J.; Kim, J. Y.; Yang, H-I.; Yoo, M. C.; Hahm, D-H.; Kim, K. S. Arthritis Res. Ther. 2009, 11:R49 DOI: 10.1186/ar2662
35. Umar, S.; Sarwar, A. H. M. G.; Umar, K.; Ahmad, N.; Sajad, M.; Ahmad, S.; Katiyar, C. K.; Khan, H. A. Cell. Immunol. 2013, 284, 51-59. DOI: /10.1016/j.cellimm.2013.07.004
36. Lu, Y.; Liu, J.; Li, H.; Gu, L. Inflammation 2016, 39, 303-308. DOI: g/10.1007/s10753-015-0250-x
37. Dolfi, F.; Pilgrim, W. R. Use of idrocilamide for the preparation of a pharmaceutical composition of rosacea. PCT WO2005/060950A1, July 7, 2005.
38. Darakhshan, S.; Pour, A. B. Pharmacol. Res. 2015, 91, 15-28 DOI: 10.1016/j.phrs.2014.10.009
39. Zhou, P.; Xiang, L.; Zhao, D.; Ren, J.; Qiu, Y.; Li, Y. Med. Chem. Commun. 2019, 10, 252-262 DOI: 10.1039/c8md00432c
40. Nissinen, L.; Kähäri, V-M. Biochim. Biophys. Acta 2014, 1840, 2571-2580 DOI: 10.1016/j.bbagen.2014.03.007
41. Chang, Y-H.; Lin, I-L.; Tsai, G. J.; Yang, S-C.; Yang, T-P.; Ho, K-T.; Tsu, T-C.; Shiau, M-Y. Clin. Biochem. 2008, 41:955-959 DOI: 10.1016/j.clinbiochem.2008.04.012
42. Li, N.; Qiao, Y.; Xue, L.; Xu, S.; Zhang, N. Int. J. Pharm. 2019, 569, 118625 DOI: 10.1016/j.ijpharm.2019.118625
43. Wajant, H.; Pfizenmaier, K.; Scheurich, P. Cell Death Differ. 2003, 10, 45-65 DOI: 10.1038/sj.cdd.4401189
44. Lipinski, C. A.; Lombardo, F.; Dominy, B. W.; Feeney, P. J. Adv. Drug Deliv. Rev. 1997, 23, 3-25 DOI: 10.1016/s0169-409x(96)00423-1
45. Shah, P.; Westwell, A. D. J. Enzyme Inhib. Med. Chem. 2007, 22, 527-540. DOI: 10.1080/14756360701425014
46. Jeffries, B.; Wang, Z.; Graton, J.; Holland, S. D.; Brind, T.; Greenwood, R. D. R.; Le Questel, J-Y.; Scott, J. S.; Chiarparin, E.; Linclau, B. J. Med. Chem. 2018, 61, 10602-10618 DOI: 10.1021/acs.jmedchem.8b01222
47. Ramírez-Marroquín, O. A.; Jiménez-Arellanes, M. A.; Cortés-Pacheco, A.; Zambrano-Vásquez, O. R.; López-Torres, A. Monatsh. Chem. 2019, 150, 267-274 DOI: 10.1007/s00706-018-2328-2
48. https://www.molinspiration.com/cgi-bin/properties , accessed in January 2020.
49. Reddy, V. M.; Nadadhur, G.; Daneluzzi, D.; Dimova, V.; Gangadharam, P. R. Antimicrob. Agents Chemother. 1995, 39, 2320-2324 DOI: 10.1128/AAC.39.10.2320
50. Philipova, I.; Valcheva, V.; Mihaylova, R.; Mateeva, M.; Doytchinova, I.; Stavrakov, G. Chem. Biol. Drug Des. 2018, 91, 763-768. DOI: 10.1111/cbdd.13140
51. Tuntiwachwuttikul, P.; Phansa, P.; Pootaeng-on, Y.; Taylor, W. C. Chem. Pharm. Bull. 2006, 54, 149-151 DOI: 10.1248/cpb.54.149
52. Sharma, S.; Kumar, M.; Sharma, S.; Nargotra, A.; Koul, S.; Khan, I. A. J. Antimicrob. Chemother. 2010, 65, 1694-1701. DOI: 10.1093/jac/dkq186
53. Sharma, S.; Kalia, N. P.; Suden, P.; Chauhan, P. S.; Kumar, M.; Ram, A. B.; Khajuria, A.; Bani, S.; Khan, I. A. Tuberculosis 2014, 94, 389-396. DOI: 10.1016/j.tube.2014.04.007
Notas de autor
*Corresponding author: Oscar Abelardo Ramírez-Marroquín, email: oramirez@unpa.edu.mx

 cygnusmind
cygnusmind