Journal of the Mexican Chemical Society, vol. 62, no. 4, 2018
Sociedad Química de México A.C.
Samira Emami
Imam Khomeini International University, Iran
Mir Mohammad Alavi Nikje *
Imam Khomeini International University, Iran
Date received: 14 August 2018
Date accepted: 29 October 2018
Abstract: In this research, the effect of (3-Aminopropyl) triethoxysilane (APTES) modified Fe3O4 (Fe3O4@APTES) core-shell nanomaterials as the recyclable heterocatalyst on the recovery of bisphenol-A (BPA) from hydrolysis of polycarbonate (PC) wastes were investigated. In the evaluated reactions, water and diethylene glycol (DEG) were used as the green solvent composition and the water as well as magnetic heterocatalyst content were optimized. By examining the results of the above-mentioned reactions, it was observed that by using 25 pbw of water and 2 pbw of magnetic heterocatalyst (both based on total waste and solvent weights), BPA achieved in 100% yield. The Fe3O4@APTES nanomaterials as the heterocatalyst can be recovered and reused up to five intervals with our significant activity losses. The resulting BPA and nanomaterials has been characterized by X-ray diffraction (XRD), and spectroscopic methods (1H NMR, 13C NMR, FT-IR).
Keywords: Bisphenol-A, Chemical recycling, Fe3O4@APTES nano-heterocatalyst, Hydrothermal condition, Hydrolysis, Polycarbonate wastes.
Resumen: En esta investigación, el efecto de (3-aminopropil) trietoxisilano (APTES) modificó los nanomateriales de Fe3O4 (Fe3O4@APTES) como heterocatalizador reciclable en la recuperación de bisfenol-A (BPA) de hidrólisis de policarbonato (PC). En las reacciones evaluadas, se utilizaron agua y dietilenglicol (DEG) como la composición del disolvente verde y se optimizó el contenido de agua y de heterocatalizador magnético. Al examinar los resultados de las reacciones mencionadas anteriormente, se observó que al usar 25 pbw de agua y 2 pbw de heterocatalizador magnético (ambos basados en residuos totales y pesos de solventes), el BPA alcanzó un rendimiento del 100%. Los nanomateriales Fe3O4@APTES como heterocatalizador pueden recuperarse y reutilizarse hasta cinco intervalos con nuestras importantes pérdidas de actividad. El BPA y los nanomateriales resultantes se han caracterizado por difracción de rayos X (XRD) y métodos espectroscópicos (1H RMN, 13C RMN, FT-IR).
Palabras clave: Bisfenol A, Reciclaje químico, Nano-heterocatalizador Fe3O4@APTES, Condición hidrotermal, Hidrólisis, Residuos de policarbonato.
Introduction
Plastics such as polycarbonate (PC) (e.g., DVDs, CDs, airplane and automobile windows, safety goggles, and computer parts) and poly (ethylene terephthalate) (PET) (e.g., PET bottles and film) made from fossil resources has been widely employed because of its high durability, excellent transparency, and good mechanical properties. Recently, the disposal of plastic products and materials has become a challenge to the achievement of sustainable societies and the development of resources recycling [1]. In the following, a number of studies on the chemical recycling of PC wastes are reviewed. Do et al. [2], has studied methanolysis of the PC into bisphenol-A (BPA) and dimethyle carbonate (DMC) using 1,5,7-triazabicyclo[(4.4.0)]-dec-5-ene (TBD) as an organocatalytic and DMC itself under mild condition. Beneš and coworkers [3], have reported the microwave-assisted degradation of the PC wastes to suitable polyols for preparing of polyurethanes (PUR) foams in optimal conditions. Depolymerization of PCs sample by the Taguchi research group to BPA was investigated using two crystallite sizes of Cerium (IV) oxide (CeO2) crystal (75 nm, and 7.40 nm) as the catalyst [1]. In another study, calcium-fuctionalized mesoporous γ-alumina (Ca-Al2O3) was used for alcoholysis of PC [4]. Of course, many other suggestions have been made by other scientists for the chemical recycling of PC wastes, from which one can refer to methods hydrolysis [5),(6),(7),(8], pyrolysis [9),(10),(11], aminolysis [12),(13], glycolysis [14),(15], and alcoholysis [16),(17),(18]. Fe3O4 is well known as the strongest magnetic nanomaterials (MNMs) of all natural minerals on earth [19]. Due to the unique features, Fe3O4 has been considered for use in a variety of applications [20),(21),(22),(23),(24),(25),(26),(27),(28]. On the other, unmodified MNMs tend to aggregate because of their high specific area and strong inter particle interaction, which limit their utilization. Therefore, it is necessary to develop strategies for the chemical stabilization of the naked MNMs against aggregation over a long period. Although the development of more efficient and versatile approaches to functionalized MNMs is very important [29]. Recently, various studies have been carried out on the use of modified Fe3O4 as the effective heterocatalyst in the synthesis of organic materials, two of which are as follows; the functionalization of silica-coated Fe3O4 magnetic nanomaterials (Fe3O4@SiO2) using chlorosulfonic acid (Fe3O4@-SiO2-SO3H) as an organic-inorganic hybrid heterocatalyst has been introduced in the one-pot synthesis of 1,8-dioxo-octahydroxanthenes derivatives [30]. In another particular case, Jafarzadeh's research group has used magnetically recoverable trifluoroacetic acid-immobilized Fe3O4@SiO2-APTES core-shell nanocatalyst for the Friedländer synthesis of quinolines under solvent-free condition [31]. Our research group has carried out in the past extensive studies on the chemical recycling of PC wastes to recover BPA as the valuable materials in the presence of sodium hydroxide (NaOH) as the homogeneous catalyst, water [32] and glycerin/water [33], and sorbitol/glycerin/water [34] under hydrothermal conditions and ethylene glycol (EG) [35] and glycerin/water [36] under microwave irradiation. Recently our research group, depolymerization of PC wastes was carried out to recover bis (4-hydroxybutyl hydrogen carbonate) of BPA (DHB-BPA), mono (4-hydroxybutyl hydrogen carbonate) of BPA (MHB-BPA) and BPA itself in the presence of (1,4-butanediol and water) as the solvent composition, nanoparticles TiO2 (NPs-TiO2) and microparticles TiO2 (MPs-TiO2) as the recyclable solid supports and NaOH as the homogeneous catalyst under simple heating method [37]. In addition, we recovery of pure BPA has been studied using hydroglycolysis of PC wastes in diethylene glycol (DEG) and water as the green solvent composition, NPs-TiO2 and MPs-TiO2 as the recyclable solid supports and NaOH as the homogeneous catalyst conditions [38]. Finally, we investigated the performance of magnetite nanomaterials coated with amine modified silica nanoshell (Fe3O4/SiO2/NH2) as an available, efficient, reusable heterocatalyst and inexpensive for recovering of BPA with yield 100% from hydrolysis of PC wastes at atmospheric pressure using water and DEG mixture without the disadvantages of traditional techniques [39]. In this research and in the continuation of previous works, our group has decided to use modified Fe3O4 as the heterocatalyst with unique features in the chemical recycling of PC wastes. Further, we have investigated the effect of magnetite nanomaterials coated with (3-Aminopropyl) triethoxysilane (APTES); as the recyclable heterocatalyst on the hydrolysis of PC wastes in order to recover pure BPA without using any homogeneous and traditional catalyst. Thus, nano-Fe3O4 coated with APTES using chemical co-precipitation method as a recyclable heterocatalyst, and using water and DEG as the green solvent composition have been used in the recovery pure BPA as valuable material of PC wastes.
Experimental
Materials
The iron trichloride hexahydrate (FeCl3.6H2O, 99%), iron dichloride tetrahydrate (FeCl2.4H2O, 99.70%), ethanol (C2H5OH, 99.70%), ammonia (NH3, 25%-28%), citric acid (C6H8O7), acetic acid (CH3COOH), APTES (97%), DEG, methanol (CH3OH), acetone (C3H6O), NaOH, and hydrogen chloride solution (HCl, 37%) bought from Merck (Darmstadt, Germany). Water for doing of reactions was twice-distilled. PC was received CDs and DVDs wastes and the thin layer of aluminum was separated completely and the large particles of PC wastes broken into the arbitrary sizes and shapes, washed with NaOH solution and hot water and dried being charged in the reactor.
Instruments
1H NMR and 13C NMR spectra were carried out using a BPUKER 300 instrument (Bruker, Billerica, Massachusetts, USA) with chloroform-d (CDCl3) as a solvent and tetramethylsilane (TMS) as an internal standard. Fourier transform infrared spectroscopy (FT-IR) spectra was done by using a BRUKER Tensor 27 (Bruker, Billerica, Massachusetts, USA) with KBr pellet press (sample/KBr mass ratio of 1:100) between 400-4000 cm-1. MNMs were characterized using an X-ray powder diffraction (XRD, Model: GNR, Novara, Italy). The XRD information was obtained on an APD-2000 diffractometer (Cu Kα radiation, λ= 1.54 A°, GNR, Novara, Italy) from 20° to 80° (°2Theta). For preparing of the heterocatalyst and drying reaction products, an ultrasonic device (Hielscher, UP200S, Oderstraße 53, 14513 Teltow, Germany), and a vacuum oven (Wisd, WOV-70, DAIHAN Scientific, Seoul, South Korea) were used, respectively. Melting point (mp) was determined on a Gallenkamp electrothermal 9100 (CO, UK).
Preparation of nano-Fe3O4
Nano-Fe3O4 were synthesized by applying previously reported conditions [40]. The nano- Fe3O4 were prepared through co-precipitation by ferric and ferrous (2/1, in moles/moles) precursors using reported method. Typically, 0.015 mol FeCl3.6H2O and 0.0075 mol FeCl2.4H2O were dissolved in twice-distilled water 1000 ml. The solution was subsequently heated to 90 °C and N2 was continuously purged in solution, for O2 exhausting. NH3 (30 ml, 25-28 %) was added drop wise and reacted for an additional 6 h with adjusting of pH around 10. The precipitate was separated by decantation and washed with ethanol and twice-distilled water. To obtain well dispersed nano-Fe3O4, the precipitate was added to citric acid (500 ml, 0.20 M) and treated with ultrasonic. Finally, unreacted acid was removed and precipitate dried in oven in 40 °C overnight.
Preparation of nano-Fe3O4@APTES
For the synthesis of nano-Fe3O4@APTES in the first step, nano-Fe3O4 (500 mg) was dispersed in 600 ml ethanol/twice-distilled water (5/1) and sonicated for 20 min by the addition of acetic acid and adjusting of pH=4. Then, 4 ml APTES was added to the solution and the mixture was stirred mechanically at room temperature for 4 h. Finally, the core-shell nanomaterial was separated and washed with twice-distilled water for four times and then dried at 45 °C in oven overnight and characterized [41] (Scheme 1).
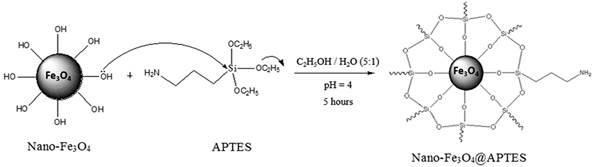
Scheme 1
Modification of nano-Fe3O4 with APTES.
General procedure for hydrolysis of PC wastes
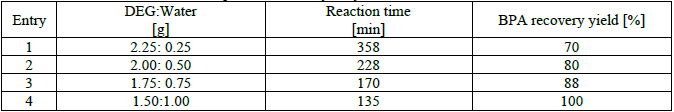
At first, DEG and water (total weights 2.50 g) as the green solvent composition, 0.08 g of nano-Fe3O4@APTES as the heterocatalyst (2 % based on total waste and solvent weights), and 1.50 g of PC wastes were introduced into a 50 ml two-necked flask and placed under reflux and magnetic stirrer. Then the reaction mixture was refluxed under atmospheric pressure and the reaction time was carefully measured until complete PC wastes. Then, the reaction mixture cooled to room temperature. In the next step, hot water and methanol were used to crystallize the BPA and nano-Fe3O4@APTES as the heterocatalyst are separated by external magnetic field. Then, HCl solution (37%) added drop wise until the BPA is completely crystallize and products are separated by filtration. In the following, washed by twice-distilled water for several times, dried at 80 °C in vacuum oven for 5 h and the obtained product was characterized. The heterocatalyst/PC wastes and total solvents/PC wastes mass ratios were 0.053 and 1.66, respectively. All results are reported in Table 1.
Effects of DEG/Water composition in the hydrolysis reaction of PC wastes.
Results and discussion
Characterization of nano-Fe3O4 and nano-Fe3O4@APTES using FT-IR, and XRD
FT-IR spectroscopy was used to confirm the correctness of the surface of the nano-Fe3O4 with APTES (Fig. 2). As can be seen, bands centered at 586 cm-1, 2918 cm-1, and 1639 cm-1 related to the Fe-O stretching vibrations (ν), νO-H, and O-H bonding vibration (σ), respectively in the FT-IR of nano-Fe3O4. Moreover, the absorption peak in the 588 cm-1 corresponding to Fe-O-Si vibrations that overlaps with the Fe-O vibrations. The band related to Si-O-Si vibrations sat 1073 cm-1 is present in the FT-IR of nano-Fe3O4@APTES. Two bands of 1748 cm-1 and 3448 cm-1 related to ν and σ of the free NH2. Finally, bands 2853 cm-1 and 2922 cm-1 are devoted to the ν of the C-H groups on the APTES.
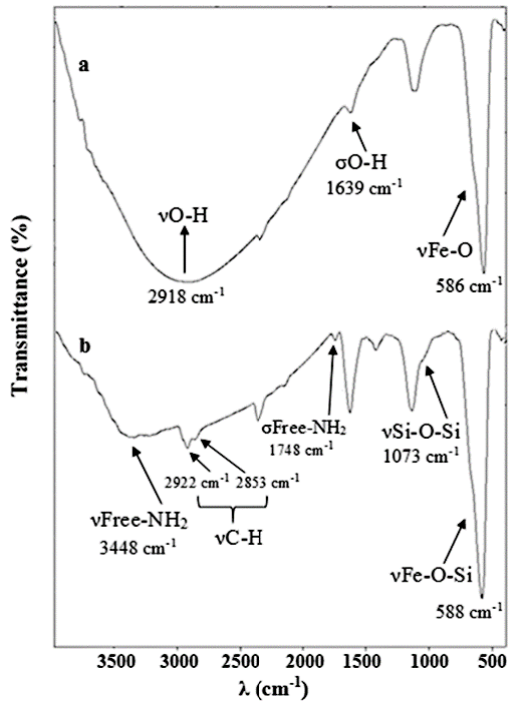
Fig. 2
FT-IR spectrums of magnetic nanomaterials; (a) nano-Fe3O4, and (b) nano-Fe3O4@APTES.
The XRD pattern of nano-Fe3O4 and nano-Fe3O4@APTES are shown in Fig. 3. The position (°2Theta) and the ratio of the intensities of all peaks in the XRD pattern of nano-Fe3O4@APTES are consistent with the XRD pattern of nano-Fe3O4, indicating the preservation of the cubic spinel structure and the non-phase change during coating of the nano-Fe3O4 [41),(42]. The average nano-Fe3O4@APTES diameter was estimated to be 29.4 nm from the XRD results with Debye-Scherrer’s equation in the °2Theta= 35.52°. Debye-Scherrer’s Eqns.1 where K is a constant (generally considered as 0.94), β is the corrected diffraction line full-width at half-maximum (FWHM), θ is Bragg’s angle and λ is the wavelength of Cu Ka (1.54 A°) [43].
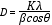 (1)
(1)
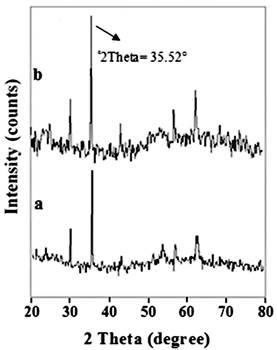
Fig. 3
XRD patterns of magnetic nanomaterials; nano-Fe3O4 (a), and nano-Fe3O4@APTES (b).
The role of solvents composition on hydrolysis of PC wastes
In order to investigate the effect of water on the hydrolysis of PC wastes, four reactions were carried out in the presence of nano-Fe3O4@APTES as the heterocatalyst, and the results are summarized in Table 1. According to the results, by increasing the amount of water in the role of the leading agent of the reaction, the reaction time is reduced and the reaction efficiency is increased.
The role of nano-Fe3O4@APTES as heterocatalyst on hydrolysis of PC wastes
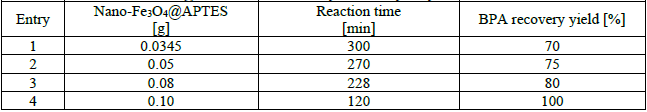
In order to investigate the effect of nano-Fe3O4@APTES as the heterocatalyst on the hydrolysis of PC wastes, four reactions were carried out in the presence of nano-Fe3O4@APTES (0.0345 to 0.10) as the heterocatalyst, DEG (2.00 g), water (0.50 g), and PC wastes (1.50 g) and the results are collected in Table 2. According to the results, with the increase for nano-Fe3O4@APTES, the reaction time is reduced and the efficiency of the reaction is increased. Moreover, with 0.10 g of nano-Fe3O4@APTES as the heterocatalyst, BPA can be obtained at 100%.
Effects of Nano-Fe3O4@APTES as heterocatalyst in the hydrolysis reaction of PC wastes.
Reusabiliting of nano-Fe3O4@APTES as the heterocatalyst
In order to recovery the nano-Fe3O4@APTES as the heterocatalyst, DEG (2.00 g) and water (0.50 g) as solvents and nano-Fe3O4@APTES (0.08 g) as the heterocatalyst, they were added to the reaction medium and 1.50 g of PC wastes was dissolved in 228 min and check the amount of PC dissolved using the method described in general procedure section. The nano-Fe3O4@APTES were extracted with the external magnetic field and washed several times with acetone and water. All these steps were repeated five times in constant time and the results are presented in Fig. 4. In the following, we compare the five times recovered nano-Fe3O4@APTES with the virgin catalyst using FT-IR and find that there are APTES coatings on nano-Fe3O4 (Fig. 5). As shown in the results, the amount of PC wastes dissolved in each period of the reaction is reduced and according to these results, the activity of the nano-Fe3O4@APTES as the heterocatalyst has decreased after five recoveries. Reducing the activity of nano-Fe3O4@APTES as the heterocatalyst may be due to a reduction in the amount of heterocatalyst in the recovery processes.
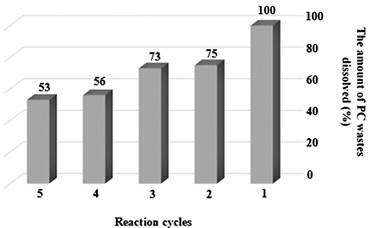
Fig. 4
Reusability of nano-Fe3O4@APTES as heterocatalyst.
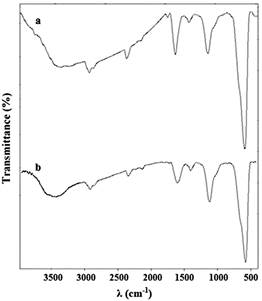
Fig. 5
FT-IR spectrums of nano-Fe3O4@APTES as heterocatalyst; (a) virgin heterocatalyst, and (b) heterocatalyst after five times reuse.
Spectroscopy studies of recovered BPA
4,4'-(propane-2,2-diyl)diphenol (BPA). White powder: mp 158°C; 1H NMR (CDCl3, 300 MHZ) δ (Fig. 6) 1.60 (6H, s, H1), 4.50 (2H, s, H2), 6.70 (4H, d, J = 8.4 Hz, H3), 7.10 (4H, d, J = 8.4 Hz, H4); 13C NMR (CDCl3, 300 MHZ) δ (Fig. 7) 114.68 (CHAr, C3), 127.95 (CHAr, C4), 143.33 (CAr-C-CH3, C5), 153.22 (C-O, C6), 31.07 (CH3, C1), 41.69 (C(CH3)2, C2); FT-IR (KBr pellet) λ (Fig. 8) 1358 cm-1 -1506 cm-1 (σC-H), 3028 cm-1 (νC-H aromatic), 2875 cm-1 (νC-H aliphatic), 3200 cm-1 to 3500 cm-1 (νO-H), 1000 cm-1 to 1300 cm-1 (νC-O), 1603 cm-1 (νC=C).
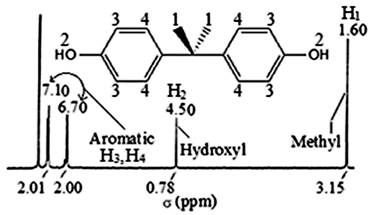
Fig. 6
1H NMR spectrum of BPA recovered.
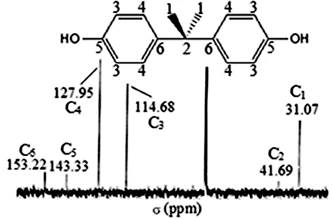
Fig. 7
13C NMR spectrum of BPA recovered.
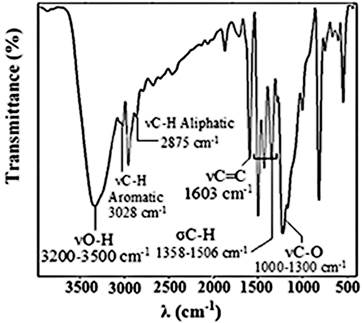
Fig. 8
FT-IR spectrum of BPA recovered.
Conclusions
In conclusion, we evaluated the nano-Fe3O4@APTES performance as the heterocatalyst in recovering of pure BPA from hydrolysis of PC wastes under atmospheric pressure in the presence of water and DEG as the green solvent composition. Under the optimal condition heterocatalyst/PC wastes and water/PC wastes where mass ratio of 0.053 and 0.66, reaction time of 135 min, pure BPA as the valuable material were recovered in 100% yield. Moreover, the Fe3O4@APTES nanomaterials as the heterocatalyst can be recovered for five cycles and reused in next reactions. Moreover, by comparing the results of the reactions, it was observed that in the presence of water (10 up 40 pbw based on total solvent weight) as the main solvent, the reaction speed and recovery of BPA increased, this corresponds to the principles of green chemistry.
Acknolwedgements
We gratefully acknowledge the information support of the Imam Khomeini International University for this research.
References
1. Taguchi, M.; Ishikawa, Y.; Kataoka, S.; Naka, T.; Funazukuri, T. Catal. Commun. 2016, 84, 93-97 DOI: https://doi.org/10.1016/j.catcom.2016.06.009+
2. Do, T.; Baral, E. R.; Kim, J. G. Polym. J. 2018, 143, 106-114 DOI: https://doi.org/10.1016/j.polymer.2018.04.015+
3. Beneš, H.; Paruzel, A.; Trhlíková, O.; Paruzel, B. Eur. Polym. J. 2017, 86, 173-187 DOI: https://doi.org/10.1016/j.eurpolymj.2016.11.030+
4. Liu, Y. Y.; Qin, G. H.; Song, X. Y.; Ding, J. W.; Liu, F. S.; Yu, S. T.; Ge, X. P. J. Taiwan Inst. Chem. Eng. 2018, 86, 222-229 DOI: https://doi.org/10.1016/j.jtice.2018.02.028+
5. Tagaya, H.; Katoh, K.; Kadokawa, J. I.; Chiba, K. Polym. Degrad. Stab. 1999, 64, 289-292 DOI: https://doi.org/10.1016/S0141-3910(98)00204-3+
6. Watanabe, M.; Matsuo, Y.; Matsushita, T.; Inomata, H.; Miyake, T.; Hironaka, K. Polym. Degrad. Stab. 2009, 94, 2157-2162 DOI: https://doi.org/10.1016/j.polymdegradstab.2009.09.010+
7. Tsintzou, G. P.; Antonakou, E. V.; Achilias, D. S. J. Hazard. Mater. 2012, 241, 137-145 DOI: https://doi.org/10.1016/j.jhazmat.2012.09.027+
8. Deirram, N.; Rahmat, A. R. APCBEE Proc. 2012, 3, 172-176 DOI: https://doi.org/10.1016/j.apcbee.2012.06.065+
9. Blazso, M. J. Anal. Appl. Pyrolysis, 1999, 51, 73-88 DOI: https://doi.org/10.1016/S0165-2370(99)00009-1+
10. Šala, M.; Kitahara, Y.; Takahashi, S.; Fujii, T. Chemosphere 2010, 78, 42-45 DOI: https://doi.org/10.1016/j.chemosphere.2009.10.036+
11. Antonakou, E. V.; Kalogiannis, K. G.; Stephanidis, S. D.; Triantafyllidis, K. S.; Lappas, A. A.; Achilias, D. S. Waste Manage. 2014, 34, 2487-2493 DOI: https://doi.org/10.1016/j.wasman.2014.08.014+
12. Hata, S.; Goto, H.; Yamada, E.; Oku, A. Polym. J. 2002, 43, 2109-2116 DOI: https://doi.org/10.1016/S0032-3861(01)00800-X+
13. Hatakeyama, K.; Kojima, T.; Funazukuri, T. J. Mater. Cycles Waste Manage. 2014, 16, 124-130 DOI: https://doi.org/10.1007/s10163-013-0151-8+
14. Li, B.; Xue, F.; Wang, J.; Ding, E.; Li, Z. Prog. Rubber Plast. Recycl. Technol. 2017, 33, 39-50.
15. Pant, D. Process Saf. Environ. Prot. 2016, 100, 281-287 DOI: https://doi.org/10.1016/j.psep.2015.12.012+
16. Hu, L. C.; Oku, A.; Yamada, E. Polym. J. 1998, 39, 3841-3485 DOI: https://doi.org/10.1016/S0032-3861(97)10298-1+
17 Liu, F.; Li, L.; Yu, S.; Lv, Z.; Ge, X. J. Hazard. Mater. 2011, 189, 249-254 DOI: https://doi.org/10.1016/j.jhazmat.2011.02.032+
18. Quaranta, E.; Sgherza, D.; Tartaro, G. Green Chem. 2017, 19, 5422-5434 DOI: 10.1039/C7GC02063E+
19. Wang, G. S.; Wang, L.; Wei, Z. Y.; Sang, L.; Dong, X. F.; Qi, M.; Chen, G. Y.; Chang, Y.; Zhang, W. X. Chin. J. Polym. Sci. 2013, 31, 1011-1021 DOI: https://doi.org/10.1007/s10118-013-1255-2+
20. Lee, D. K.; Kang, Y. S.; Lee, C. S.; Stroeve, P. J. Phys. Chem. B 2002, 106, 7267-7271 DOI: https://pubs.acs.org/doi/abs/10.1021/jp014446t+
21. Liao, M. H.; Chen, D. H. J. Mater. Chem., 2002, 12, 3654-3659 DOI: 10.1039/B207158D+
22. Yang, T.; Shen, C.; Li, Z.; Zhang, H.; Xiao, C.; Chen, S.; Xu, Z.; Shi, D.; Li, J.; Gao, H. J. Phys. Chem. B, 2005, 109, 23233-23236 DOI: https://pubs.acs.org/doi/abs/10.1021/jp054291f+
23. Tan, S. T.; Wendorff, J. H.; Pietzonka, C.; Jia, Z. H.; Wang, G. Q. Chem. Phys. Chem., 2005, 6, 1461-1465 DOI: https://onlinelibrary.wiley.com/doi/abs/10.1002/cphc.200500167+
24. Yang, H. H.; Zhang, S. Q.; Chen, X. L.; Zhuang, Z. X.; Xu, J. G.; Wang, X. R. J. Anal. Chem. 2004, 76, 1316-1321 DOI: https://pubs.acs.org/doi/abs/10.1021/ac034920m+
25. Zhang, D.; Liu, Z.; Han, S.; Tang, T., Liu, X.; Han, S.; Lei, B., Zhou, C. Nano Lett. 2004, 4, 2151-2155 DOI: https://pubs.acs.org/doi/abs/10.1021/nl048758u+
26. Sun, S.; Murray, C. B.; Weller, D.; Folks, L.; Moser, A. Science 2000, 287, 1989-1992 DOI: 10.1126/science.287.5460.1989+
27. Frias, J. C.; Ma, Y.; Williams, K. J.; Fayad, Z. A.; Fisher, E. A., Nano Lett. 2006, 6, 2220-2224 DOI: https://pubs.acs.org/doi/abs/10.1021/nl061498r+
28. Caruntu, D.; Caruntu, G.; Chen, Y.; O'Connor, C. J.; Goloverda, G.; Kolesnichenko, V. L. Chem. Mater. 2004, 16, 5527-5534 DOI: https://pubs.acs.org/doi/abs/10.1021/cm0487977+
29. Yang, X.; Chen, L.; Han, B.; Yang, X.; Duan, H. Polym. J. 2010, 51, 2533-2539 DOI: https://doi.org/10.1016/j.polymer.2010.04.032+
30. Naeimi, H.; Nazifi, Z. S. J. Nanopart. Res. 2013, 15, 2026-2037 DOI: https://doi.org/10.1007/s11051-013-2026-2+
31. Jafarzadeh, M.; Soleimani, E.; Norouzi, P.; Adnan, R.; Sepahvand, H. J. Fluorine Chem. 2015, 178, 219-224 DOI: https://doi.org/10.1016/j.jfluchem.2015.08.007+
32. Alavi Nikje, M. M.; Askarzadeh, M. Prog. Rubber Plast. Recycl. Technol. 2014, 30, 145-152.
33. Alavi Nikje, M. M.; Askarzadeh, M. Polimery W. 2013, 23, 29-31 DOI: http://dx.doi.org/10.1590/S0104-14282013005000019+
34. Alavi Nikje, M. M.; Askarzadeh, Polimery W. 2013, 58, 292-294 DOI: dx.doi.org/10.14314/polimery.2013.292+
35. Alavi Nikje, M. M. Polimery W. 2011, 56, 381-384.
36. Alavi Nikje, M. M.; Askarzadeh, M. Prog. Rubber Plast. Recycl. Technol. 2013, 29, 169-176.
37. Emami, S.; Alavi Nikje, M. M. Iran. Polym. J. 2018, 27, 275-286 DOI: https://doi.org/10.1007/s13726-018-0607-8+
38. Emami, S.; Alavi Nikje, M. M. Green Process Synth. in press DOI: https://doi.org/10.1515/gps-2018-0028+
39. Emami, S.; Alavi Nikje, M. M. Russ. J. Appl. Chem. 2018, 91, 159-166 DOI: https://doi.org/10.1134/S107042721801024X+
40. Alavi Nikje, M. M.; Nejad, M. A.; Shabani, K.; Haghshenas, M. Colloid Polym. Sci. 2013, 291, 903-909 DOI: https://doi.org/10.1007/s00396-012-2808-6+
41. Alavi Nikje, M. M.; Moghaddam, S. T.; Noruzian, M.; Farahmand Nejad, M. A; Shabani, K.; Haghshenas, M.; Shakhesi, S. Colloid Polym. Sci. 2014, 292, 627-633 DOI: https://doi.org/10.1007/s00396-013-3099-2+
42. Saif, B.; Wang, C.; Chuan, D.; Shuang, S. J. Biomater. Nanobiotechnol. 2015, 6, 267-275 DOI: http://dx.doi.org/10.4236/jbnb.2015.64025+
43. Massart, R. IEEE trans. Magn. 1981, 17, 1247-1248 DOI: 10.1109/TMAG.1981.1061188+
Author notes
*Corresponding author: Mir Mohammad Alavi Nikje, e-mail address: Drmm.alavi@gmail.com

 cygnusmind
cygnusmind