Journal of the Mexican Chemical Society, vol. 62, no. 3, 2018
Sociedad Química de México A.C.
Héctor A. Peniche-Pavía
Centro de Investigación Científica de Yucatán, Calle 43 #130, Col. Chuburná de Hidalgo, Mérida, Yucatán, México 97205., México
Marina Vera-Ku
Centro de Investigación Científica de Yucatán, Calle 43 #130, Col. Chuburná de Hidalgo, Mérida, Yucatán, México 97205., México
Sergio R. Peraza-Sánchez *
Centro de Investigación Científica de Yucatán, Calle 43 #130, Col. Chuburná de Hidalgo, Mérida, Yucatán, México 97205., México
Date received: 15 November 2017
Date accepted: 19 February 2018
Funding
Funding source: National Council of Science and Technology of Mexico (Conacyt)
Contract number: #105346
Funding
Funding source: Conacyt
Contract number: #338183
Abstract: The main objective of this review is to update the current knowledge on species of Dorstenia genus. Previous review was published in 2000. This genus includes 117 accepted named species, from which metabolites with a great variety of structures are found. In the last fifteen years, a blooming has occurred in the research of the genus, with more than 60 original research papers on the chemistry and pharmacology of many species, being one of the main reasons to this thriving the generalized use of Dorstenia species in the traditional medicine around the world.
Keywords: Phytochemistry and Pharmacology, Dorstenia, coumarins, terpenes, flavonoids.
Resumen: El objetivo principal de esta revisión es actualizar los conocimientos existentes sobre especies del género Dorstenia. Una revisión previa fue publicada en 2000. Este género incluye 117 especies aceptadas, las cuales se encuentran metabolitos con una gran variedad de estructuras. En los últimos quince años se ha producido un gran auge en la investigación del género, con más de 60 trabajos de investigación originales sobre la química y farmacología de muchas especies, siendo una de las principales razones para este florecimiento el uso generalizado de especies de Dorstenia en la medicina tradicional en todo el mundo.
Palabras clave: Fitoquímica y Farmacología, Dorstenia, cumarinas, terpenos, flavonoides.
Introduction
The purpose of the present review is to update the previous review article on Dorstenia genus by Abegaz et al. and the review done in a book chapter by Ngadjui & Abegaz [1,2]. Both works contributed in collecting the first phytochemical and pharmacological information on species of this genus, mentioning the study on the genus by Casagrande et al. [3], in which the structure of syriogenin is reported from D. contrajerva, along with the most recent data on D. elliptica, D. zenkeri, and D. ciliata not yet published at that moment.
For this work, four databases (or search engines) were used: CONRICYT metasearch engine, Google Scholar, PubMed, and SciFinder. According to CONRICYT metasearch engine, the total number of publications is 596 in the period 2000-2016 using the term Dorstenia in some part of the text; in Google Scholar, 2,510; in PubMed, 2,451; and in SciFinder, 121. The results showed that the vast majority of articles found were not relevant to the subject of the review, so the search was restricted to a more precise selection, establishing the following search and selection criteria: a) advanced search by title and in abstract with the term Dorstenia; b) articles only related on the pharmacology of species of Dorstenia, natural products isolated from them, and the biological activity of the latter; c) the publication time lapse included only articles from 2000 to 2016; d) the search was performed in English, but some articles found in Portuguese and in Spanish were also included.
The number of articles found applying such search criteria are as follows: for Google Scholar 101, for the metasearch engine of CONRICYT 158, from PubMed 50, and from SciFinder 31. This search allowed us to have a comprehensive understanding of the genus. However most of the papers found were related to the botanical description of Dorstenia species, so they had to be excluded leaving us with a total of 66 papers for study.
The genus Dorstenia includes 345 described species and 117 accepted named species and is widely distributed from Central to South America (the New World), Africa, and even Asia (the Old World) [4]; since a great variety of metabolites are found in this genus, we have described the principal and characteristic compounds of this genus in different sections.
In the last fifteen years, a blooming in the research of the genus occurred, with more than 60 original research papers on the chemistry of diverse species; one of the main reasons to this thriving is the generalized use of Dorstenia species in traditional medicine around the world. For example, in Brazil, an extract called “carapia” obtained from the roots of the endemic species D. brasiliensis, D. arifolia, and D. multiformis is used to cure diseases related to skin problems [5, 6], whereas in Cameroon Dorstenia species are used to cure a diverse array of human disorders, including arthritis, rheumatism, gout, stomach ailments, malaria, cough, headache, and skin diseases [7].
We divided this review into four sections, the first one includes coumarin and furanocoumarin compounds and their prenylated forms; the second section covers prenylated flavonoids, in particular, 2-(benzyl)-benzofuran compounds, a family of metabolites that has only appeared in this genus; a third section is dedicated to steroids, triterpenes, and other lipids; in addition, we are dedicating the last section to the most recent biological studies related to Dorstenia species and their compounds.
Phytochemistry
Coumarins and furanocoumarins. This set of metabolites has been studied in recent years by the group of Dr. Jürgen Schmidt, who has been dedicated to the phytochemistry on plants of Yemen. This led them to work on the isolation of new 15 furanocoumarins from leaves and twigs of D. gigas [8] and leaves of D. foetida [9] The former one gave the compounds swietenocoumarin B (5), swietenocoumarin F (6), furanopinnarin (7), dimethoxychalpensin (12), isoimperatorin (19), cnidilin (20), oxypeucedanin (21), byakangelicin (32), other six monoprenylated derivatives of psoralen (8-10, 13, 14), a monogenarylated derivative of psoralen (33), and six angelicin derivatives (34-39). From the latter one, the isolation process yielded psoralen (1), isopimpinellin (4), swietenocoumarin (5), 5-methoxychalepensin (11), turbinatocoumarin (15), malonylturbinatocoumarin (16), phellopterin (31) and other two prenylated compounds (17, 18), plus five derivatives of chalepensin (22-26).
In Africa, the medicinal plants of Cameroon have been actively studied by the Department of Organic Chemistry, University of Yaonde I, in Yaounde, Cameroon. The newly studied species and discovered compounds include: coumarin and bergapten (3) from the leaves and twigs of D. picta [10]; twigs of D. prorepens showed the two linear furanocoumarins psoralen (1) and bergapten (3) [11]; and from the twigs of D. elliptica the new compounds 29 and 30 were isolated plus the known compounds dorstenin (28), bergapten (3), and psoralen (1) [12].
From the twigs of D. turbinata turbinatocoumarin (15) was isolated for the first time plus two known furanocoumarins, 1 and 13 [13]. From the twigs of D. dinklagei, which yielded principally triterpenes (Section 3), the common bergapten (3) was found too [14] (Fig. 1). However, it is more common to see in reports on Dorstenia species a greater variety of either prenylated flavonoids or sterols and triterpenes than furanocoumarins, particularly in African Dorstenia species.
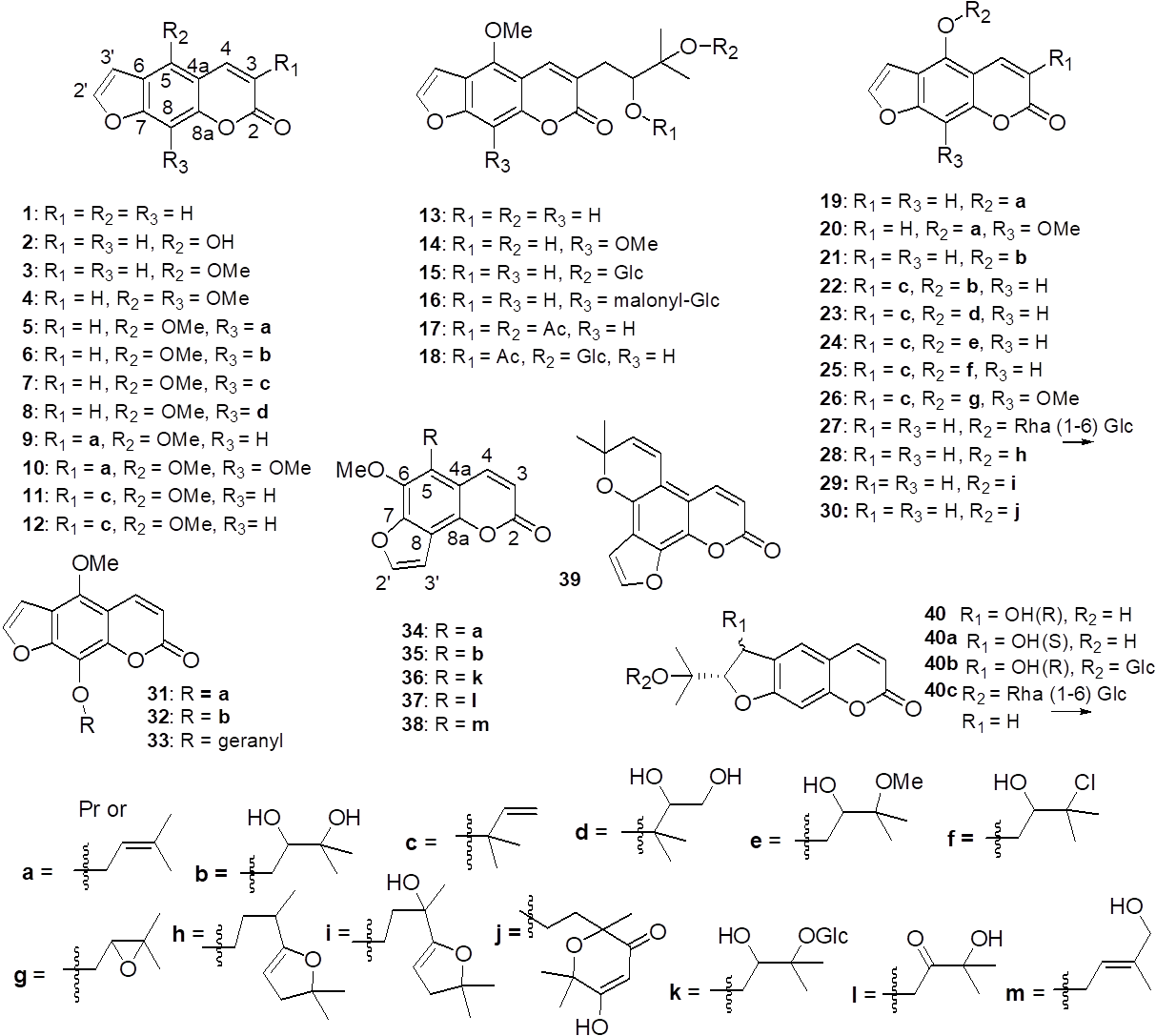
Fig. 1
Furanocoumarins isolated from species belonging to the genus Dorstenia.
About the recent research in the Americas, in Central America the aerial parts of D. contrajerva afforded a glycosylated furanocoumarin, α-L-rhamnopyranosyl-(1→6)-β-D-glucopyranosyl-bergaptol, plus compounds 1-3 and 27 [15]. In Brazil, an analysis with HPLC showed D. tubicina and D. asaroides containing psoralen (1) and bergapten (2) only in the rhizomes, whereas D. vitifolia showed solely isopimpinellin (4) in both rhizomes and aerial parts [16]. Other species studied from Brazil, such as D. multiformis, yielded psoralen (1) and bergapten (3) [5], the rhizomes of D. bahiensis yielded bergapten (3) and dorstenin (28), while in D. arifolia psoralen (1) was isolated [17], so this showed that linear furanocoumarins substituted in position 5 are the most common of coumarins reported from Dorstenia species of the New World.
The isolation of hydrofuranocoumarins on Dorstenia species is not that common as the furanocoumarins. Therefore, only a few of these compounds has been reported in recent years within the genus, smyrindiol (40) from D. brasiliensis and D. turbinata, two glycosylated derivatives of smyrindiol (40b & 40c) isolated from D. brasiliensis, and xanthoarnol (40a) from D. contrajerva [13,18,19].
Prenylated flavonoids. A great number of prenylated flavonoids from African Dorstenia species has been identified in a short period of time; sometimes one species can show a lot of similarity between the compounds isolated from another species. These compounds are mono-, di- and triprenylated flavonoids, which include flavans, chalcones, flavones, flavonones and flavonols, and dimers of flavonoids (Figures 2 and 3).
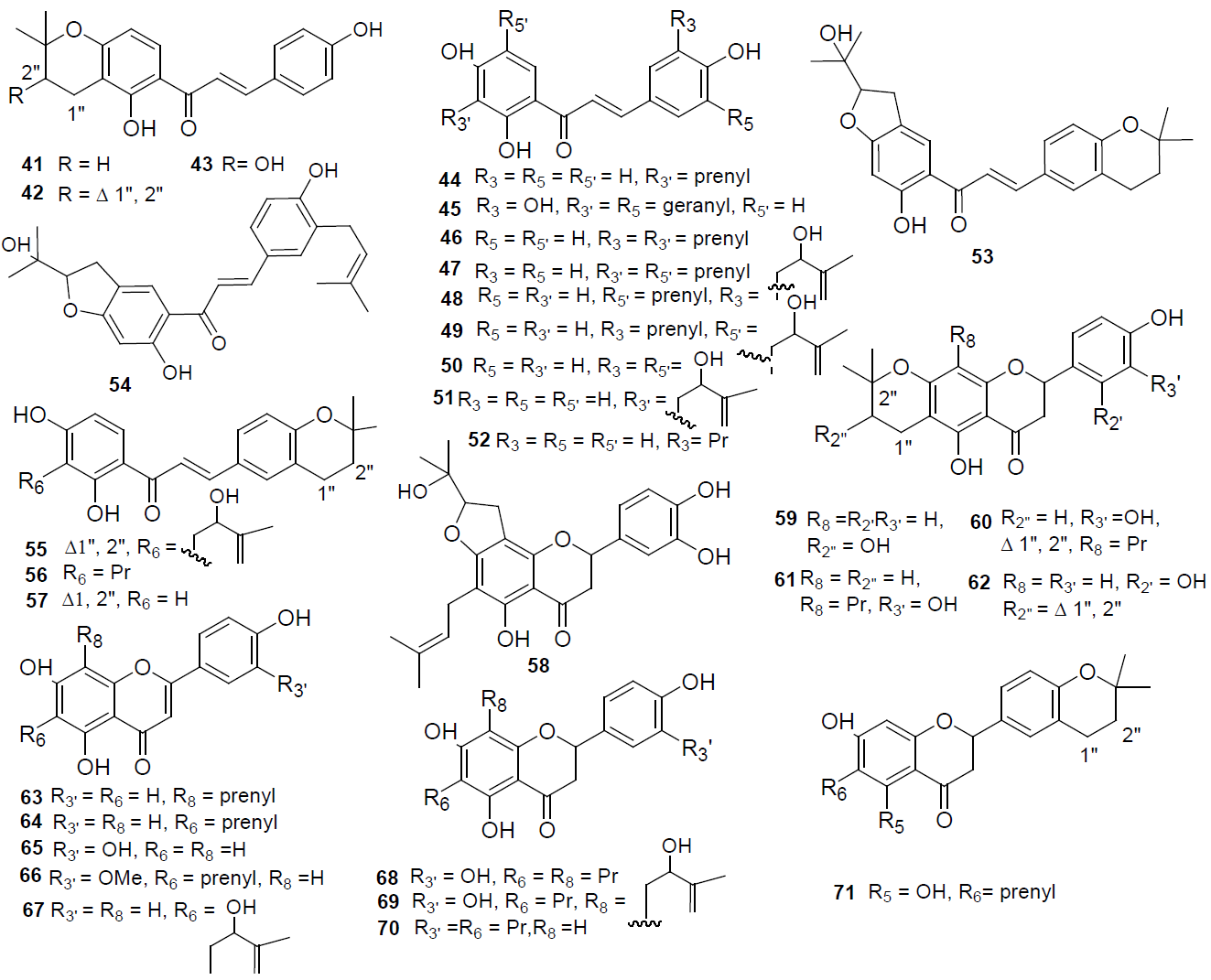
Fig. 2
Prenylated flavonoids isolated from Dorstenia species.

Fig. 3
Prenylated flavonoids and flavonoid dimers isolated from Dorstenia species.
The best example to first begin this part of the review is the study of prenylated compounds on the aerial parts of D. mannii [20-22]. In this species alone eleven compounds named after the plant have been reported, the monoprenylated chalcone dorsmanin A (41) and the diprenylated flavonoids dorsmanin B (76), C (77), D (80), E (74), F (58), G (72), H (69), I (60), and J (61), 6-prenylchrysoeriol (64), and the important 6,8-diprenyleriodictyol (68) from which it is possible the biosynthesis of the dorsmanins C to J, so 6,8-diprenyleriodictyol (68) is a key compound for the chemical diversity in D. mannii [1].
From Dorstenia species growing in Cameroon numerous new compounds have been yielded. In the twigs of D. dinkanglei three new compounds were found: dinklagin A (71), a diprenylated flavanone, and the monoprenylated flavones dinklagin B (59) and dinklagin C (67) [23]. Also, the new bis geranylated chalcone prorepensin (45) was isolated from the twigs of D. prorepens. Meanwhile, in the aerial parts of D. zenkeri a new monoprenylated chalcone (43) was found along with the dimer known as dorstenone (90) [11], the latter formed by Diels-Alder reaction between the bridge chain of a first chalcone and a prenyl moiety annexed to ring A of a second chalcone, which made it different to the dimer 88 isolated from D. barteri var. multiradiata, which has a prenyl moiety attached to ring A as a pyrane ring on the second chalcone, forming a bicyclic fused ring on the structure of the dimer [24].
Other species have been marketed to be used as medicinal plants, such as D. ciliata, which yielded from the aerial parts two new prenylated flavones, ciliatins A (73) and B (85) [14], as well as isobavachalcone (44), stipulin (47), 8-prenylapigenin (63), 6-prenylapigenin (64), luteolin (65), canniflavone (66), dinklagin C (67), and poinsettifolin A (84); and from the twigs of D. elliptica the 3-(prenyl)-4,2’,4’-trihydroxychalcone (52) and a new flavan (83), the only known isolated from this genus [12].
Meanwhile, the twigs of D. barteri var. subtriangularis showed four new prenylated chalcones, named as bartericins A (48), B (54), C (53), and D (49), plus stipulin (47), kanzonol B (57), and 3’-(2-hydroxy-3-methylbut-3-enyl)-4,2’,4’-trihydroxychalcone (51) [25,26]; if we compare them with the compounds isolated from D. barteri var. multiradiata, these prenylated chalcones do not form any heterocyclic ring with the prenyl chain, such as in isobavachalcone (44), stipulin, kanzonol C (46), and 4-hydroxylonchocarpin (42), albeit from this species the bis-flavonoid amenthoflavone (89) has been isolated. Kanzonol C (46) and 4-hydroxylonchocarpin (42) were also found in the twigs of the Cameroonian D. turbinate [27]. Other three new prenylated chalcones were found in the twigs of D. angusticornis, which were named as angusticornin A (55), B (50), and C (91), plus stipulin (47), gancaonin Q (70), paratocarpin C (56), and paratocarpin F (79) [26, 28].
The last report on prenylated compounds from species of Dorstenia in the African continent that we are reviewing is that of the species D. psilurus, which has a great amount of triprenylated flavonols, including six new dorsilurins: F (81), G (82), H (87), I (78), J (86), and K (75). These dorsilurins are C-4’ prenylated but not hydroxylated, as normally occurs with other flavonoids reviewed in this paper [29].
To the best of our knowledge, there is only one case of a prenylated flavonoid isolated from a Dorstenia species of the New World, cycloartocarpesin (62), which was isolated from D. contrajerva [19]. This is very interesting because it demonstrates that prenylated flavonoids could also be found in Dorstenia species native to the Americas.
From Dorstenia species, 2-benzyl-benzofuran compounds have been obtained (Fig. 4), and until now, to the best of our knowledge, three 2-benzyl-benzofurans have only been isolated in nature, in particular, from species of Dorstenia genus. The first one (92) was obtained from the twigs of D. gigas [8], an endemic plant from Yemen. A total synthesis was reported for this compound, describing and confirming its structure [30]. The second and third compounds were named as dorsjervin A (93) and dorsjervin B (94), both having been isolated from D. contrajerva, a native plant from Central America [19]. It was proposed aurones as biosynthetic precursors of 2-benzyl-benzofurans and also as chemotaxonomic markers for this genus.
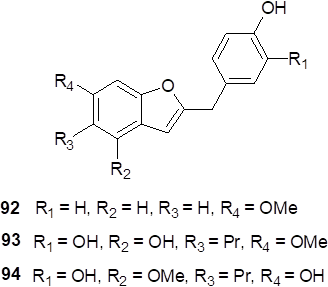
Fig. 4
2-Benzyl-benzofurans isolated from Dorstenia species.
Sterols, pentacyclictriterpenes, and other lipids. Normally, phytosterols such as β-sitosterol (131) and pentacyclic triterpenes such as α-amyrin (97) and β-amyrin (98) and their esters and glycoside derivatives are ubiquitous in the genus Dorstenia and are the main types of lipids reported (Figures 5 and 6). Also, some glycolipids and diterpenes have been isolated.

Fig. 5
Pentacyclic triterpenes and sterols found in Dorstenia species.

Fig. 6
Pentacyclic triterpenes, sterols and other lipids found in Dorstenia species.
In the New World, reports based on research done in South America, in particular with native species from Brazil, have shown that D. arifolia contains a great variety of ester derivatives of α- and β-amyrin (95-108), 11-oxo-α-amyrin and 11-oxo-β-amyrin (109-118), plus glutinol (119), glutinyl acetate (120), ursa-9(11),12-dien-3-yl acetate (121), oleana-9(11),12-dien-3-yl acetate (122), ursa-9(11),12-dien-3-yl decanoate (123), oleana-9(11),12-dien-3-yl decanoate (124), 12,13-epoxyolean-3-yl acetate (125), 12,13-epoxyolean-9(11)-en-3-yl acetate (126), taraxeryl acetate (127), lupenyl acetate (128), lanosta-8,24-dien-3-yl acetate (129), but no new compounds were isolated [31]. Another native species from Brazil, D. multiformis, also yielded α-amyrin acetate (95) and β-amyrin acetate (96), although no new compounds were found in this species [5]. In another work, roots of D. brasiliensis showed the capacity of producing two new seco-adiananes: dorstenic acids A (134) and B (135) plus the only known diterpene in the genus, 14α-hydroxy-7,15-isopimaradien-18-oic acid (149) [18]. Meanwhile, in Central America from the rhizomes of D. contrajerva γ-sitosterol (132) and the monogalactosyl-diacylglycerol (18:0/18:3) (148) were found [19].
In Cameroon, studies on endemic Dorstenia plants showed they have not only prenylated flavonoids and furanocoumarins but some triterpenes. From the twigs of D. dinklagei α-amyrenonyl acetate (110), 3β-acetoxy-1β,11α-dihydroxy-olean-12-ene (137), α-amyrin acetate (96), and the new compound β-acetoxy-6α-hydroxy-11-oxoisobauerene (136), named as dinklagenonoate [32], were isolated. On the other hand, twigs and air-dried leaves of D. picta yielded three known compounds: β-sitosterol (131), β-sitosterol glucopyranoside (133), and oleanolic acid (130) [10].
Meanwhile, D. convexa yielded a good variety of lupane triterpenes, still no one of them were new compounds, but it was the first isolation from the genus Dorstenia of betulonic acid (139), platanic acid (140), betulinic acid (141), 3,20-dioxo-30-norlupan-28-oic acid (142), lupenone (143), and 29-norlupan-3,20-dione (144), plus the ubiquitous lupeol (138) and the sterol β-sitosterol glucopyranoside (132), previously isolated from this genus [33]. As happened with D. arifolia , in D. harmsiana long chain alkanoic acid esters were present too, but lupeol (138) and their derivatives were also found, the new compounds lup-20(29)-en-3-nonanoate (145) and lup-20(29)-en-3-undecanoate (146), and the known compound lup-20(29)-en-3-hexadecanoate (147) [34].
An interesting molecule was found in the roots of D. picta, another species from Cameroon. This compound was named dorstenpictanona (150), and this is the first report found in this genus about a bicyclic polyprenylated compound [35].
This part of the review permits to get an insight in forthcoming phytochemical and metabolomic studies on the genus Dorstenia and the possible answer we will get if we do investigate species of this genus without previous phytochemical research. Therefore, if we choose to study a new species of Dorstenia we can expect non-polar fractions to yield principally sterols and triterpenes and their ester derivatives, while in the more polar fractions we can expect to obtain furanocoumarins, in particular linear furanocoumarins and their derivatives substituted principally in C-5, besides prenylated flavonoids.
Pharmacological activities of extracts and pure compounds from Dorstenia species
In the last fifteen years, the pharmacological properties of a lot of Dorstenia species have been studied for the first time. Normally, the biological activity data are first obtained for the extracts analyzed in a panel of bioassays, and later the pure compounds isolated from those extracts are tested, but some studies directly describe the biological activity of pure compounds without previous research on the extracts.
The two principal bioactivities tested for the extracts of Dorstenia species are related to antioxidant and biocidal effects. The antioxidant effect has been studied in recent years on D. barteri, D. convexa, D. foetida, D. psilurus, and D. ciliata. Generally, these extracts show good or moderate activities in the DPPH test; this antioxidant effect is related to the presence of flavonoids and other phenolic compounds in the extracts [36-40]. Concerning the microbicidal effect, research on antifungal and antibacterial activities has been reported for D. barteri Bureau var. multiradiata, D. elliptica, D. contrajerva, D. mannii, and D. angusticornis on a panel of fungi, and Gram positive and Gram negative bacteria cultures, respectively. But the best results were found on extracts of twigs of D. barteri var. multiradiata, which showed moderate activity with a MIC of 19.5 μg/mL in Gram negative bacteria culture [27, 41-46].
The antiprotozoal effect is evident for the methanol extract of D. contrajerva rhizomes against Giardia lamblia, Leishmania mexicana, L. tropica, and L. major [47-49]. The methanol extract of D. barnimiana was tested against Plasmodium falciparum, L. infantum, Trypanosoma cruzi, T. brucei, and MRC-5, a human lung normal cell line, the results showing that D. barnimiana had moderate activity (IC50 = 29.6-49.4 μg/mL), but not selectivity [50,51]. Against P. falciparum, fractions of the methanol extract of D. convexa twigs had a good activity (IC50 < 5 μg/mL) [52]. Interestingly, the methanol extract of D. klaineana had moderate activity (IC50 = 17 μg/mL) against P. falciparum, but a strong cytotoxicity on MRC-5 (IC50 = 0.43 μg/mL) [53]. The extract of D. barteri var. multiradiata twigs was active against Trichomonas gallinarum [39].
The extracts from D. psilurus, D. gigas, D mannii, and D. elliptica have been evaluated on cytotoxic assays and for induction of apoptosis properties. Only the extract from D. psilurus had good activities with IC50’s from 7.18 to 18.0 μg/mL in five lines of human promyelocytic leukemia cells, pancreatic cancer cells, and prostate cancer cells, plus the results showed that extract of D. psilurus induced apoptosis through mitochondria related mechanisms on HL-60 cells [54-56].
Other activities are more focused on the physiological effects that the extracts can produce; thus, for the nervous central system, sedative and anticonvulsant activities were found on the methanol extract of D. arifolia rhizome in mice, which may be related to a facilitation of the GABAergic transmission [57]. D. barteri var. subtriangularis twigs extract possesses both anti-inflammatory and antinociceptive activities, confirmed by various chemical-, mechanical-, and thermal-induced pain test models and acute carrageenan-induced paw edema [58]. D. gigas leaves chloroform extract showed a 65% inhibitory capacity of acetylcholinesterase, an Alzheimer disease drug target, at a concentration of 0.2 mg/mL, comparable to the reference drug galanthamine [59].
The extract of D. psilurus twigs showed potential antihypertensive effects on hyperinsulinemic rats, preventing and reversing high blood pressure, induced by a diet rich in fructose, probably by improvement of plasma insulin and cholesterol levels [60]. On the other hand, the methanol extract of D. picta twigs showed in vivo and in vitro anti-natriuretic activity and demonstrated antidiabetic effect in in vivo experiments on streptozotocin-induced diabetic rats [61, 62]. The aqueous extract of the aerial parts of D. contrajerva showed better results in comparison to loperamide on the inhibition of intestinal secretion response to cholera toxin test [63].
Finally, an experiment was done to assess the cicatrizing effect of an ointment made with the grinded rhizomes of D. drakena, an endemic species from Mexico, showing better cicatrizing effect than the positive control Recoverón®, a commercial brand with the cicatrizing agent sodium acexamate [64].
One of the first studies committed to test the activity of isolated metabolites, rather than studying the whole extract, reported the activity of three flavonoids from D. mannii, a plant used to treat rheumatism and stomach disorder: 6,8-diprenyleriodictyol (68), dorsmanin C (77), and dorsmanin F (58), which showed potent inhibition of LDL oxidation by induction of Cu2+ (IC50 < 1.0 μM) that can be relevant to the treatment or prevention of atherosclerosis, and these three compounds had better results than butylated hydroxytoluene (BHT) in DPPH test [65].
The triprenylated flavonoids dorsilurins F (81), G (82), H (87), I (78), J (86), and K (75) were examined for their potential use in diabetes treatment, but exhibited moderate to low α-glucosidase inhibitory activity [20].
Studies on the antitrichromonal and antioxidant activities of prenylated flavonoids isolated from the CH2Cl2/MeOH (1:1) extracts of D. barteri and D. convexa presented results for antioxidant activity in the DPPH assay expressed in EC50 (µg/mL) = ascorbic acid (positive control, 19.33) < 6,8-diprenyleriodictyol (68) (32.12) < bartericin A (48) (47.85) < dorsmanin F (58) (53.89) < isobavalchalcone (44) (84.33). Meanwhile, the antitrichomonal activity showed the minimum lethal concentration (MLC, µg/mL) after 24 h for each compound: quercitrin (positive control, 0.12) > bartericin B (54) (0.24) > bartericin A (48) (0.73) > 6,8-diprenyleriodictyol (68) = isobavachalcone (44) = dorsmanin F (58) (31.25) [39].
Two studies done to discover the potential antibacterial activity of pure compounds isolated from D. barteri var. multiradiata and D. angusticornis versus a panel of more than 20 microbial cultures confirmed that isobavachalcone (44), angusticornin B (50) and bartericin A (48) possess excellent in vitro activity in the macrodilution assay in comparison to the reference drugs, gentamycin for the Gram positive and Gram negative bacteria and nystatin for yeast [27, 46].
Continuing on the microbicidal research, the report of Dzoyem et al. (2013) showed the activity and mechanism of action of four prenylated flavonoids, 6,8-diprenyleriodictyol (68), yielded from the aerial parts of D. mannii, isobavachalcone (44) and 4-hydroxylonchocarpin (42), isolated from the twigs of D. barteri, and 6-prenylapigenin (64), from the twigs of D. dinklagei, against eight strains of MRSA (methicillin-resistant Staphylococcus aureus). The results showed 6,8-diprenyleriodictyol (MIC = 0.5-4.0 μg/mL) to be more active, followed by isobavachalcone (MIC = 0.5-16.0 μg/mL) and 4-hydroxylonchocarpin (MIC = 1-8 μg/mL), while 6-prenylapigenin (MIC = 16-64 μg/mL) being the least active. According to the authors, the bactericidal effect is related to the capacity of these prenylated flavonoids to produce damage on the bacterial membrane, which includes depolarization of the membrane, disrupting transport of nutrients, therefore, halting biosynthetic processes that lead to cell death [66].
The same four flavonoids were also tested for anti-inflammatory and anticholinesterase activity, having 6,8-diprenyleriodictyol (68) had the best result on NO inhibition of 90.71% at a concentration of 3.12 μg/mL, while isobavachalcone (44) showed the best results of inhibition of 15-lipoxygenase and anticholinesterase with IC50’s of 25.92 and 5.93 μg/mL, respectively [67].
The CH2Cl2/CH3OH (1:1) extract from D. barteri var. multiradiata and their isolated flavonoids isobavachalcone (44), kanzonol C (46), 4-hydroxylonchocarpin (42), stipulin (47), and amentoflavone (89) were tested for antimycobacterial, antigonorrheal and anti-reverse transcriptase activities (anti-VIH). The anti-reverse transcriptase activity was significantly lower for all the samples analyzed than the reference compound doxorubicin, however, isobavachalcone did it well or even better in comparison to the reference drug used in the antimycobacterial (ciprofloxacin and isoniazid) and antigonorrheal (gentamycin) tests, with MIC’s less than 10 μg/mL for all the strains tested [68].
Research on antiprotozoal activity includes pure antileishmanial compounds, such as poinsettifolin A isolated from D. poinsettifolia, which showed great activity against promastigotes of L. braziliensis and L. amazonensis with IC50’s = 2.5 and 4.9 µg/mL, respectively [69]. From D. contrajerva cycloartocarpesin (62) and galactolipid (149) had a good activity against promastigotes of L. mexicana (IC50 = 4.9 and 2.9 μM, respectively) [19]. The search for antimalarial activity on the isolated compounds from D. barteri var. subtriangularis showed the potential of three prenylated chalcones: bartericin A (48), stipulin (47), and 4-hydroxylonchocarpin (42) [70].
The anticancer potential of secondary metabolites isolated from Dorstenia species has also been studied multiple times. The prenylated flavonoids kanzonol C (46), 4-hydroxylonchocarpin (42), isobavachalcone (44), paratocarpin C (56), stipulin (47) and dorsmanin A (41) of D. barteri var. multiradiata [71] and D. turbinata [72] showed that were even better than the two reference compounds (chlorogenic acid and epigallocatechin-3-gallate) to inhibit matrix metalloproteinase (MMP-2) from U87 glioblastoma cells. The expression of MMP-2 is correlated to the degradation of the extracellular matrix that permits the movement of cancer cells to other tissues in the process called metastasis.
A new study on the anticancer potential with fourteen cancer cell lines and a normal hepatic cell (AML-12) of common flavonoids of Dorstenia showed gancaonin Q (70), 6-prenylapigenin (64), 6,8-diprenyleriodictyol (68), and 4-hydroxylonchocarpin (42) to have good activity, in particular, gancaonin Q and 4-hydroxylonchocarpin showed to be ‘hits’ for four cancer cell lines. In the in vitro cytotoxic assay, it is accepted as good result when compounds show CC50’s < 10 μM, being those compounds named as ‘hits’. The mechanism implicated in the cytotoxic effect was activation of caspases 3/7 on CCRF-CEM cells [73].
We encountered a tendency to investigate the activity of prenylated flavonoids more than in any of the other groups of metabolites found in Dorstenia. However, examples of other compounds can be found in the literature, such as furanocoumarins. Thus, compounds 13 and 15 isolated from D. turbinata showed low activity against a panel of bacterial and fungi pathogens, except against the fungus Trichophyton rubrum, where their activity was notable (MIC = 9.76 μg/mL) [13]. The furanocoumarins have been thoroughly studied, in particular for their use in phototherapy applied on skin diseases, for instance, psoriasis, eczema, vitiligo, and others [74].
Conclusions
The number of publications about Dorstenia had a peak in 2010-2011, but afterwards it started going downhill as seen in Figure 7, probably due to the fact that contemporary papers are more focused to botanical description of lesser known species of the genus Dorstenia, a necessary step to get deeper phytochemical or biological activity knowledge later. Another trend important to note is that the quantity of papers related to isolation and elucidation of natural products was greater in the first years than the pharmacology related ones. This is due to the fact that more pharmacology research was done after more knowledge about the chemical constitution was obtained first, and those compounds isolated through phytochemical research were tested for different biological activities, giving in total a proportion of two phytochemical research papers for each three pharmacological activity papers. This tendency nicely shows how the applied science depends on basic science.
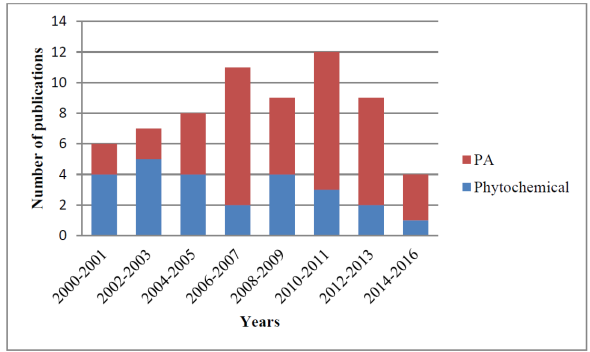
Fig. 7
Number of publications related to the pharmacological activity (PA) and phytochemical research completed on Dorstenia species.
This review shows that there is a great difference of groups of metabolites encountered in species of Dorstenia from Africa, in which prenylated flavonoids are predominant, in comparison with species from the Americas and Asia, with a greater proportion of lipids and furanocoumarins. However, phytochemical studies tend to show only the more abundant compounds; therefore, these studies do not give complete information of all the compounds present in the extracts, because most of the compounds are present on small proportions; also, it is common for researchers to specialize on the direct isolation to a certain class of compounds. Thus, differences between species are due in part to the distinct techniques, methodologies, and objectives of each research used in the articles reviewed here and not just on the expected variations among the metabolomic profiles of Dorstenia species, coming from their own genomic diversity between the genus’ species [75].
Currently, we have plentiful studies related to the biocidal capacities of the extracts and pure compounds from Dorstenia, tested on in vitro and murine model assays. However, we could not find data related to clinical studies to confirm the efficacy and non-toxic effects on humans of some extracts that have showed true potential use to the treatment or prevention of the diseases studied in the papers reviewed. The studies undertaken on pure compounds, such as the monoprenylated flavonoids bartericin A (48) and isobavachalcone (44), showed to have bigger potential to be developed as new drugs than the rest of the natural products in the Dorstenia genus. These monoprenylated flavonoids deserve more research, including in vivo experimental assays and in silico studies to optimize their structures, which are necessary steps to find the best drug-like compound to be introduced to the clinical stages of drug development.
Acknowledgements
We thank National Council of Science and Technology of Mexico (Conacyt) for research grant #105346. HAPP thanks Conacyt for scholarship #338183.
References
1. Abegaz, B. M.; Ngadjui, B. T.; Dongo, E. Curr. Org. Chem. 2000, 4, 1079-1090.
2. Ngadjui, B. V. T.; Abegaz, B. M. In Studies in Natural Products Chemistry (Part J); 2003; pp 761-805.
3. Casagrande, C.; Ronchetti, F.; Russo, G. Tetrahedron 1974, 30, 3587-3589.
4. The Plant List (2013). Version 1.1. Published on Internet; Published on Internet; http://www.theplantlist.org/1.1/browse/A/Moraceae/Dorstenia/ (accessed Sep 2, 2017).
5. Balestrin, L.; Gaspari Dias, J. F.; Miguel, O. G.; Dall’Stella, D. S. G.; Miguel, M. D. Brazilian J. Pharmacogn. 2008, 18, 230-235.
6. Celeghini, R. M. S.; Yariwake, J. H.; Lanças, F. M. Rev. Bras. Plantas Med. 2007, 9, 61-66.
7. Kuete, V.; Efferth, T. Front. Pharmacol. 2010, 1.
8. Franke, K.; Porzel, A.; Masaoud, M.; Adam, G.; Schmidt, J. Phytochemistry 2001, 56, 611-621.
9. Heinke, R.; Franke, K.; Porzel, A.; Wessjohann, L. A.; Awadh Ali, N. A.; Schmidt, J. Phytochemistry 2011, 72, 929-934.
10. Vouffo, B.; Hussain, H.; Eyong, K. O.; Dongo, E.; Folefoc, G. N.; Nkengfack, A. E.; Krohn, K. Biochem. Syst. Ecol. 2008, 36, 730-732.
11. Abegaz, B. M.; Ngadjui, B. T.; Dongo, E.; Ngameni, B.; Nindi, M. N.; Bezabih, M. Phytochemistry 2002, 59, 877-883.
12. Abegaz, B. M.; Ngadjui, B. T.; Folefoc, G. N.; Fotso, S.; Ambassa, P.; Bezabih, M.; Dongo, E.; Rise, F.; Petersen, D. Phytochemistry 2004, 65, 221-226.
13. Ngameni, B.; Kuete, V.; Simo, I. K.; Mbaveng, A. T.; Awoussong, P. K.; Patnam, R.; Roy, R.; Ngadjui, B. T. South African J. Bot. 2009, 75, 256-261.
14. Ngadjui, B. T.; Dongo, E.; Abegaz, B. M.; Fotso, S.; Tamboue, H. Phytochemistry 2002, 61, 99-104.
15. Caceres, A.; Rastrelli, L.; De Simone, F.; De Martino, G.; Saturnino, C.; Saturnino, P.; Aquino, R. Fitoterapia 2001, 72, 376-381.
16. Cardoso, C. A. L.; Vilegas, W.; Barison, A.; Honda, N. K. J. Agric. Food Chem. 2002, 50, 1465-1469.
17. Lopes, D.; Oliveira, R. R.; Kaplan, M. A. C.; Lage, C. S.; Leitão, A. C. Planta Med. 2001, 67, 820-824.
18. Uchiyama, T.; Hara, S.; Makino, M.; Fujimoto, Y. Phytochemistry 2002, 60, 761-764.
19. Peniche-Pavía, H. A.; Medrano-Nahuat, D.; Torres-Tapia, L. W.; Mut-Martín, M.; García-Miss, R.; Peraza-Sánchez, S. R. Phytochem. Lett. 2016, 18, 140-143.
20. Ngadjui, B. T.; Abegaz, B. M.; Dongo, E.; Tamboue, H.; Fogue, K. Phytochemistry 1998, 48, 349-354.
21. Ngadjui, B. T.; Dongo, E.; Tamboue, H.; Fogue, K.; Abegaz, B. M. Phytochemistry 1999, 50, 1401-1406.
22. Ngadjui, B. T.; Kouam, S. F.; Dongo, E.; Kapche, G. W. F.; Abegaz, B. M. Phytochemistry 2000, 55, 915-919.
23. Ngadjui, B. T.; Ngameni, B.; Dongo, E.; Kouam, S. F.; Abegaz, B. M. Bull. Chem. Soc. Ethiop. 2002, 16, 157-163.
24. Tsopmo, A.; Tene, M.; Kamnaing, P.; Ayafor, J. F.; Sterner, O. J. Nat. Prod. 1999, 62, 1432-1434.
25. Ngameni, B.; Ngadjui, B. T.; Folefoc, G. N.; Watchueng, J.; Abegaz, B. M. Phytochemistry 2004, 65, 427-432.
26. Ngadjui, B. T.; Watchueng, J.; Keumedjio, F.; Ngameni, B.; Simo, I. K.; Abegaz, B. M. Phytochemistry 2005, 66, 687-692.
27. Mbaveng, A. T.; Ngameni, B.; Kuete, V.; Simo, I. K.; Ambassa, P.; Roy, R.; Bezabih, M.; Etoa, F. X.; Ngadjui, B. T.; Abegaz, B. M.; Meyer, J. J. M.; Lall, N.; Beng, V. P. J. Ethnopharmacol. 2008, 116, 483-489.
28. Simo, I. K.; Ngadjui, B. T.; Abegaz, B. M. Biochem. Syst. Ecol. 2005, 33, 1283-1287.
29. Tabopda, T. K.; Ngoupayo, J.; Awoussong, P. K.; Mitaine-Offer, A. C.; Ali, M. S.; Ngadjui, B. T.; Lacaille-Dubois, M. A. J. Nat. Prod. 2008, 71, 2068-2072.
30. Correa, C.; Cruz, M. D. C.; Jiḿnez, F.; Zepeda, L. G.; Tamariz, J. Aust. J. Chem. 2008, 61, 991-999.
31. Fingolo, C. E.; De S. Santos, T.; Filho, M. D. M. V.; Kaplan, M. A. C. Molecules 2013, 18, 4247-4256.
32. Vouffo, B.; Krohn, K.; Kouam, S. F.; Hussain, H.; Dongo, E.; Meier, K.; Schulz, B. Biochem. Syst. Ecol. 2008, 36, 655-658.
33. Poumale, H. M.; Awoussong, K. P.; Randrianasolo, R.; Simo, C. C.; Ngadjui, B. T.; Shiono, Y. Nat Prod Res 2012, 26, 749-755.
34. Poumale, H. M. P.; Amadou, D.; Shiono, Y.; Kapche, G. D. W. F.; Ngadjui, B. T. Asian J. Chem. 2011, 23, 525-527.
35. Hussain, H.; Vouffo, B.; Dongo, E.; Riaz, M.; Krohn, K. J. Asian Nat. Prod. Res. 2011, 13, 547-550.
36. Abdou-Bouba, A.; Njintang, Y. N.; Scher, J.; Mbofung, C. M. F. Agric. Biol. J. North Am. 2010, 1, 213-224.
37. Etoundi, C. B.; Kuaté, D.; Ngondi, J. L.; Oben, J. J. Nat. Prod. 2010, 3, 165-171.
38. Kansci, G.; Dongo, E.; Genot, C. Nahrung - Food 2003, 47, 434-437.
39. Omisore, N. O. A.; Adewunmi, C. O.; Iwalewa, E. O.; Ngadjui, B. T.; Adenowo, T. K.; Abegaz, B. M.; Ojewole, J. A.; Watchueng, J. Brazilian J. Med. Biol. Res. 2005, 38, 1087-1094.
40. Marwah, R. G; Fatope, M. O.; Al-Mahrooqi, R.; Varma, G.; Al-Abadi, H.; Al-Burtamani, S. S. S. Food Chem. 2007, 101,465-470
41. Alanís, A. D.; Calzada, F.; Cervantes, J. A.; Torres, J.; Ceballos, G. M. J. Ethnopharmacol. 2005, 100 , 153-157.
42. Kechia, F. A.; Gerald, N. T.; Ngameni, B.; Fokunang, C.; Dzoyem, J. P.; Kamga, H. L. African J. Biotechnol. 2016, 15, 62-67.
43. D’Angelis, C. E. M.; Leite, M. F.; Sousa, J. P. B.; Alonso, L.; Polizello, A. C. M.; Groppo, M.; Aires, C. P.; Bastos, J. K.; Spadaro, A. C. C. Anaerobe 2012, 18, 31-36.
44. Fokunang, C. N.; Mejane, N. .; Noumie, E.; Ngameni, B.; Gonsu, H. .; Kechia, F. .; Awoussong, K. P.; Guedje, N. M.; Iofack, R.; Fokunang-Tembe, E. A.; Ngoupayo, J.; Ndikum, V.; Tabi, O. Y.; Ngadjui, B. T. J. Microbiol. Antimicrob. 2012, 4, 88-95.
45. Kuete, V.; Ngameni, B.; Tsafack, A. M.; Ambassa, P.; Simo, I. K.; Bezabih, M.; Etoa, F. X.; Ngadjui, B. T.; Abegaz, B. M.; Beng, V. P. Pharmacologyonline 2007, 1, 573-580.
46. Kuete, V.; Simo, I. K.; Ngameni, B.; Bigoga, J. D.; Watchueng, J.; Kapguep, R. N.; Etoa, F. X.; Tchaleu, B. N.; Beng, V. P. J. Ethnopharmacol. 2007, 112, 271-277.
47. Peraza-Sánchez, S. R.; Cen-Pacheco, F.; Noh-Chimal, A.; May-Pat, F.; Simá-Polanco, P.; Dumonteil, E.; García-Miss, M. R.; Mut-Martín, M. Fitoterapia 2007, 78, 315-318.
48. Peraza-Sánchez, S. R.; Poot-Kantún, S.; Torres-Tapia, L. W.; May-Pat, F.; Simá-Polanco, P.; Cedillo-Rivera, R. Pharm. Biol. 2005, 43, 594-598.
49. Getti, G.; Durgadoss, P.; Domínguez-Carmona, D.; Martin-Quintal, Z.; Peraza-Sánchez, S.; Peña-Rodriguez, L. M.; Humber, D. J. Parasitol. 2009, 95, 456-460.
50. Mothana, R. A.; Al-Musayeib, N. M.; Matheeussen, A.; Cos, P.; Maes, L. Molecules 2012, 17, 14349-14360.
51. Al-Musayeib, N. M.; Mothana, R. A.; Matheeussen, A.; Cos, P.; Maes, L. BMC Complement. Altern. Med. 2012, 12, 1123.
52. Boyom, F. F.; Kemgne, E. M.; Tepongning, R.; Ngouana, V.; Mbacham, W. F.; Tsamo, E.; Zollo, P. H. A.; Gut, J.; Rosenthal, P. J. J. Ethnopharmacol. 2009, 123, 483-488.
53. Lekana-Douki, J. B.; Bongui, J. B.; Oyegue Liabagui, S. L.; Zang Edou, S. E.; Zatra, R.; Bisvigou, U.; Druilhe, P.; Lebibi, J.; Toure Ndouo, F. S.; Kombila, M. J. Ethnopharmacol. 2011, 133, 1103-1108.
54. Kuete, V.; Krusche, B.; Youns, M.; Voukeng, I.; Fankam, A. G.; Tankeo, S.; Lacmata, S.; Efferth, T. J. Ethnopharmacol. 2011, 134, 803-812.
55. Mothana, R. A. A.; Grünert, R.; Lindequist, U.; Bednarski, P. J. Pharmazie 2007, 62, 305-307.
56. Pieme, C. A.; Guru, S. K.; Ambassa, P.; Kumar, S.; Ngameni, B.; Ngogang, J. Y.; Bhushan, S.; Saxena, A. K. BMC Complement. Altern. Med. 2013, 13, 223.
57. Zapata-Sudo, G.; Mendes, T. C. F.; Kartnaller, M. A.; Fortes, T. O.; Freitas, N. F. B.; Kaplan, M. A. C.; Sudo, R. T. J. Ethnopharmacol. 2010, 130, 9-12.
58. Omisore, N. O. A.; Adewunmi, C. O.; Iwalewa, E. O.; Ngadjui, B. T.; Watchueng, J.; Abegaz, B. M.; Ojewole, J. A. O. J. Ethnopharmacol. 2004, 95, 7-12.
59. Bakthir, H.; Ali, N. A. A.; Arnold, N.; Teichert, A.; Wessjohann, L. Afr J Tradit Complement Altern Med. 2011, 8, 296-299.
60. Dimo, T.; Rakotonirina, a; Tan, P. V; Dongo, E.; Dongmo, a B.; Kamtchouing, P.; Azay, J.; Abegaz, B. M.; Cros, G.; Ngadjui, T. B. Phytomedicine 2001, 8, 101-106.
61. Ngueguim-Tsofack, F.; Dimo, T.; Kouambou, N. C.; Tchamadeu, M.-C.; Dzeufiet, D. P. D.; Tedong, I.; Renaud, B.; Kamtchouing, P. Pharmacologyonline 2006, 3, 900-911.
62. Ngueguim-Tsofack, F.; Markadieu, N.; Crutzen, R.; Donfack, J. H.; Dongo, E.; Kamtchouing, P.; Dimo, T.; Beauwens, R. Pak. J. Pharm. Sci. 2013, 26, 537-545.
63. Velázquez, C.; Calzada, F.; Torres, J.; González, F.; Ceballos, G. J. Ethnopharmacol. 2006, 103, 66-70.
64. Méndez-Martínez, G. M.; Montalvo-Javé, E. E.; Toledo, E. W.; Fernández, R. O.; Enriqueta, A.; Franco, B. Cir. Gen. 2008, 30, 204-210.
65. Dufall, K. G.; Ngadjui, B. T.; Simeon, K. F.; Abegaz, B. M.; Croft, K. D. J. Ethnopharmacol. 2003, 87, 67-72.
66. Dzoyem, J. P.; Hamamoto, H.; Ngameni, B.; Ngadjui, B. T.; Sekimizu, K. Drug Discov. Ther. 2013, 7, 66-72.
67. Dzoyem, J. P.; Nkuete, A. H. L.; Ngameni, B.; Eloff, J. N. Arch. Pharm. Res. 2015.
68. Kuete, V.; Ngameni, B.; Mbaveng, A. T.; Ngadjui, B.; Meyer, J. J. M.; Lall, N. Acta Trop. 2010, 116, 100-104.
69. Escobar, Z.; Solano, C.; Larsson, R.; Johansson, M.; Salamanca, E.; Gimenez, A.; Muñoz, E.; Sterner, O. Tetrahedron 2014, 70, 9052-9056.
70. Ngameni, B.; Watchueng, J.; Boyom, F. F.; Keumedjio, F.; Ngadjui, B. T.; Gut, J.; Abegazd, B. M.; Rosenthalc, and P. J. Arkivoc 2007, 13, 116-123.
71. Ngameni, B. ; Touaibia, M.; Patnam, R.; Belkaid, A.; Sonna, P.; Ngadjui, B. T.; Annabi, B.; Roy, R. Arkivoc 2007, 9, 91-103.
72. Ngameni, B.; Touaibia, M.; Patnam, R.; Belkaid, A.; Sonna, P.; Ngadjui, B. T.; Annabi, B.; Roy, R. Phytochemistry 2006, 67, 2573-2579.
73. Kuete, V.; Ngameni, B.; Wiench, B.; Krusche, B.; Horwedel, C.; Ngadjui, B. T.; Efferth, T. Planta Med. 2011, 77, 1984-1989.
74. Prabhu, S.; Shenoi, S. Indian J. Dermatology, Venereol. Leprol. 2014, 80, 497.
75. Misiewicz, T. M.; Zerega, N. C. Edinburgh J. Bot. 2012, 69, 413-440.
Author notes
* Corresponding author: S.R. Peraza-Sánchez Tel. (999) 942-8330, Ext. 264, e-mail: speraza@cicy.mx

 cygnusmind
cygnusmind