Journal of the Mexican Chemical Society, vol. 61, no. 3, 2017
Sociedad Química de México A.C.
Farhad Salimi *
Kermanshah Branch, Islamic Azad University, Iran
Keivan Tahmasobi
Kermanshah Branch, Islamic Azad University, Iran
Changiz Karami
Kermanshah Branch, Islamic Azad University, Iran
Alireza Jahangiri
Shahrekord University, Iran
Date received: 03 February 2017
Date accepted: 16 May 2017
Abstract: Modified nano-silica with Bismuth and Iron adsorbent was synthesized to be used as an effective adsorbent material for methylene blue (MB) removal from water solution. The prepared samples were characterized using SEM, FTIR, XRD and TEM. The effect of experimental parameters such as pH, contact time and initial concentration on adsorption treatment were studied. Results indicated that the optimum conditions for maximum adsorption of 20 mg/L MB were: contact time of 20 minutes, pH= 5-6 and 8 gr/L adsorbent, the remaining MB in solution was 1.75%. Langmuir and Freundlich isotherms were employed to model the experimental results and the Freundlich isotherm was the best-fitting models for the experiment results. The kinetic data were also analyzed through pseudo-first-order and pseudo-second-order models. The pseudo-second-order kinetic model well depicted the kinetics of dyes adsorption on adsorbent.
Key words: adsorption, modified nano- silica with bismuth and iron, bismuth, iron, methylene blue.
Resumen: Nano-silice modificado con bismuto y hierro se evaluó como adsorbente de azul de metileno (MB) presente en solución acuosa. Los adsorbentes se caracterizaron por SEM, FTIR, XRD y TEM. El pH, tiempo de contacto y concentración inicial de MB fueron algunas variables estudiadas en el proceso de adsorción. Para una adsorción máxima de 20 mg/L MB se encontraron las siguientes condiciones óptimas: 20 minutos de tiempo, pH= 5-6 y 8 gr/L de adsorbente. Los resultados de adsorción se ajustaron al modelo de Freundlich mientras que la cinética de la tinta en el adsorbente se describe mediante un modelo de pseudo segundo orden.
Palabras clave: adsorción, nano-silice, bismuto, hierro, azul de metileno.
1. Introduction
Each year millions of tons of colored materials are discharged through the wastewater of various industries such as plastic, leather, textile, paper and paint manufacturers, etc. Dye is one of the most dangerous groups of chemical compounds found in industrial wastewaters. These materials cause allergies, skin irritation [(1)], and cancer for human beings[(2)] and they also disturb the photosynthesis process[(3)]. Methylene blue is one of the most dangerous dyes. If one smells methylene blue, they suffer from breathing disorder and if the eye of animals or human beings has the contact with this material, it would be permanently harmed and they will suffer from nausea as well [(4, 5)]. There are many methods for removing dyes from industrial wastewaters such as coagulation[(6)]،, chemical oxidation[(7)], biological treatment[(8)] and membrane processes [(9)], etc. Many of these common technologies have their own economic limitations. Therefore, it is necessary to present some solutions regarding the costs of the methods so that the effectiveness of the process would not be reduced.
One of the most efficient methods to remove dyes from wastewaters is the adsorption, because it is simple, its investment does not cost so much and it is not sensitive to toxic substances. Moreover, it is rather easy to revive the adsorbent after the adsorption for it to be reused in the adsorption process[(10)].
Silica gel is one of the nanoparticles which is commonly used as a catalyst and an adsorbent. Nano-silica with a diameter of 30 nanometers is prepared using the sol-gel method which is common. Recently, Nano-Iron metals have been added to Nano-silica and it is now used to remove the pollutants through the Fenton reaction[(11)]. Amorphous surface properties of silica, which is considered as an oxide adsorbent, depends on the presence of Silanol groups (OH) in many cases. OH groups act as the molecular adsorption centers during the interaction with the adsorbate. In this case, hydrogen bonds can be formed with the OH groups or it totally undergoes the donor-acceptor interaction[(12)].
At pH greater than its iso-electric point (>2), silica exhibits ion exchange capabilities via the weakly aciditic silanol groups [(13)] and can be used to adsorb various types of inorganic [(14)] and organic [(15)] contaminant in water. The silica-encapsulated magnetite which was prepared by sol-gel method has been reported for desulfurization from natural gas [(16)] and adsorption of methyl orange[(17)], zinc [(18)] and uranium [(19)] from aqueous solution.
The adsorption behaviors of typical anionic (red Congo) and cationic (methylene blue) dyes on modified silica gel sorbent were examined by Sheikhian. The results indicated that the most percent adsorption of RC molecules were related to acidic pH values, while basic pH values were more effective than acidic ones for adsorption of MB molecules [(20)]. Anbia et al. carried out batch sorption experiments to remove dyes from aqueous solutions using a mesoporous silica SBA-3 as an adsorbent. Results indicated that the uncalcined SBA-3 adsorbent has a large adsorption capacity and a strong affinity for the anionic dyes[(15)]. Han et al. (2016) prepared Thehydrophobic silica aerogel (MSA) and hydrophilic silica aerogel (HAS) by ambient pressure drying method and examined the ability of removal of Rhodamine B (RhB) and Methylene blue (MB) from aqueous solution. Results showed that the maximum adsorptions of MB were 65.74 mg/g, 47.21 mg/g for MSA and HSA, respectively. The maximum adsorptions of RhB were 134.25 mg/g and 185.61 mg/g for MSA and HSA, respectively[(21)]. Kai Ma et al. prepared PEGylatedmesoporous silica nanoparticles with a single pore, tunable size around 9 nm and narrow size distributions that can be labeled with near-infrared dye Cy5.5[(22)]. Kakarla et al. used a new strategy for synthesis of hybrid nanocomposites based on silanized magnetite nanoparticles and sulfonatedpolyanilines[(23)]. Yu ping et al. synthesized SiO2/Poly (3-aminophenylboronic) composites by NaF and D-fructose in the polymerization medium[(24)]. To date, various adsorbents have been used for removal of dye pollutions from the aqueous solution which can be found in previous literatures [(21, 25-33)].
Bimetallic nanoparticles have attracted enormous attention in the past decade. They provide exciting opportunities for developing novel catalysts with unique or improved activities [(34)]. In addition, one of the bimetallic catalyst heavy post-transition elements like bismuth, lead, or thallium with transition elements like Pd- or Pt was used for the selective organic reaction such as oxidation of alcohols into aldehydes and carboxylic acids and catalytic oxidation of glucose [(35)]. The promoting role of other post-transition elements like Te or transition metals for reactions has also been investigated [(36)]
In the present work, modified SiO2 were prepared through precipitated iron and bismuth on nano SiO2. The characterization of prepared sample was accomplished using various instruments, such as, powder X-ray diffraction (XRD), FTIR (Fourier transform infrared), transmission electron microscopy (TEM), and SEM (Scanning Electron Microscope). The prepared modified silica were examined to adsorb methylene blue in water
2-Materials and methods
2-1-Materials
Silicon oxide (SiOx), with a diameter of 15 nanometers was bought from the Nanostructured and Amorphous Materials Company and iron nitrate (Fe(NO3)2.5H2O with a purity of 99%), bismuth nitrate (Bi(NO3)3·5H2O with a purity of 99.99%), potassium hydroxide (KOH with a purity of 99.99%), sodium carbonate (NaOH with a purity of 99.99%) and other materials such as methylene blue with high purity were bought from Merck Company. In the present study, methylene blue is a compound of hydrochloric acid, nitric acid and double distilled water.
2-2-Preparation method of Modified Nano-SiO2 by Bismuth/Iron
Firstly, all of the required containers were washed and dried using hydrochloric acid and nitric acid with a 1/3 ratio. 2 g of Nano-silica gel were dissolved in 50mL of distilled water in a 100-mL flask. Then, 15% of the weight of the iron nitrate (0.3g of this powder) was stirred on the heater stirrer along with 15% of nitrate bismuth (0.3g) so that we would have a homogeneous solution. After that 20% v/w sodium bicarbonate was slowly added to this solution and litmus paper was used to monitor the pH to reach 10. Next, the solution was heated until the temperature reached 100ºC. At this stage, the solution was kept in that condition for 15 minutes and then it was refluxed for 5 to 6 hours. After that, the solution cooled down and the obtained precipitation was put in an electric furnace at 700 ºC for three hours. At this point, the solution goes through filtration and is washed a few times with double distilled water to separate Na+.([(37, 38)].
2-3-Instruments
UV-vis absorption spectra were acquired on a Cary 100 UV-vis spectrometer (Varian, USA) at room temperature (23-25 °C) by using a double beam. FT-IR spectra were measured on a bruker spectrophotometer pressed into KBr pellets and is reported in wave numbers (cm-1). Transmission electron microscopy (TEM) was carried out on a PHILIPS CM30- 200KVand FESEM images were obtained using a HITACHI S-4160 field emission scanning electron microscope. X-ray powder diffraction (XRD) analysis has been carried out on a BRUKER B8 ADVANCE X-ray diffractometer with CuKα radiation. Typically, a scanning velocity of 1.5°∙min-1 was used to scan the peaks of the adsorbent diffraction pattern in the 2θ range between 5 and 80°.
A Metrohm 692 pH meter (Herisau, Switzerland) and Scanning Electron Microscope (Cam Scan MV2300) were used for pH measurements and nanoparticles size, respectively.
2-4-Method
In this study, different experiments were performed in order to investigate the adsorption of dyes such as methylene blue. These experiments are: 1- adsorbing methylene blue using Nano- silica and 2- adsorbing methylene blue using Nano-silica gel modified by iron and bismuth. In order to perform the second experiment, given the type of the experiment and the study, a contact was made between the methylene blue solutions at the required concentrations with a specific amount of the adsorbent (Nano-silica gel modified by iron and bismuth) at various pHs. Then, the solution was stirred for a certain amount of time. The resulting solution was poured into a test tube and then it was placed in centrifuge at a 4000 rpm speed for 20 minutes so that modified Nano-silica gel by iron and bismuth would remain at the bottom of the test tube. Ultimately, the amount of adsorption of the resulting solution was calculated using spectrophotometer. Percentage of adsorption is calculated using the following equation.
In this equation, Cf and Ci are the initial and final concentrations of methylene blue in the solution, respectively. The methylene blue with a purity of 99% used to preapare the solution has been bought from Merck Company in Germany. Various concentrations of methylene blue have been used to prepare the 5 to 100ppm solution and the amount of adsorption was reviewed with 0.08g of Nano-boehmite and bismuth. Then, the following equation was used to determine the adsorption capacity.
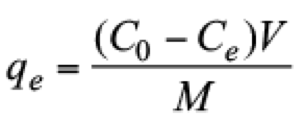 (2)
(2)In this equation, qe is the adsorption capacity, C0 and Ce are the initial and the equilibrium concentrations of the dye in the liquid phase (mg/L), respectively. V is the volume of the solution (L) and M is the amount of adsorbent used in this experiment (g).
3-Results and Discussion
Here the structure of the adsorbents is analyzed using SEM, TEM, XRD and FTIR.Accordingly, the effect of various factors such as pH, contact time, amount of adsorbent and so on will be investigated. Ultimately, the efficiency of the adsorbents for purifying methylene blue will be shown.
3-1-Investigation of absorbents’ structure using FTIR
Fig. 1 shows the FT-IR spectrum of nano-SiO2. The IR band at 3450 cm-1 is due to the stretching vibration of H2O molecules. Correspondingly, the IR band at 1632 cm-1 is due to the bending vibration of H2O molecules. The shoulder at 3246 cm-1 could be assigned to the stretching vibrations of Si-OH groups in the structure of amorphous SiO2. The presence of the Si-OH group is proved as bonded water.
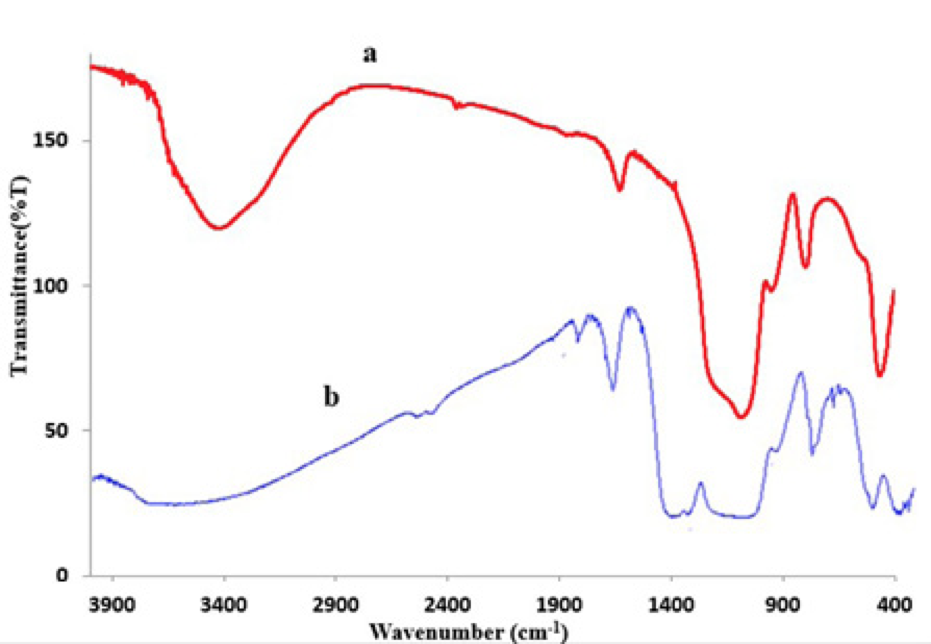
Figure 1
a) the FTIR image of Nano-silica gel b) Nano-silica gel modified by iron and bismuth.
The very strong and broad IR band at 1000 cm-1 is usually assigned to the TO and LO modes of the Si-O-Si asymmetric stretching vibrations. The IR band at 956 cm-1 can be assigned to silanol groups. In the case of alkali silicate glasses, this band is assigned to Si-O- stretching vibrations. The IR band at 800 cm-1 can be assigned to Si-O-Si symmetric stretching vibrations, whereas the IR band at 474 cm-1 is due to O-Si-O bending vibrations [(21, 39)]. Figure 1b which illustrates Nano-silica gel modified by iron and bismuth is similar to the figure above. The only difference between these two is that the Bi-O trend peaks at 1100 cm-1[(40)] and the Fe trend reaches a peak at 660 cm-1 which is an indication of the presence of bismuth and iron in the structure[(41)].
3-2-Investigating the structure of adsorbents using SEM
The SEM image associated with Nano-silica gel modified by iron and bismuth have been presented in figure 2a. As the figure illustrates, Nano-silica gel modified by iron and bismuth is a rod. It is seen on the surface of the particles indicating the presence of iron and bismuth.
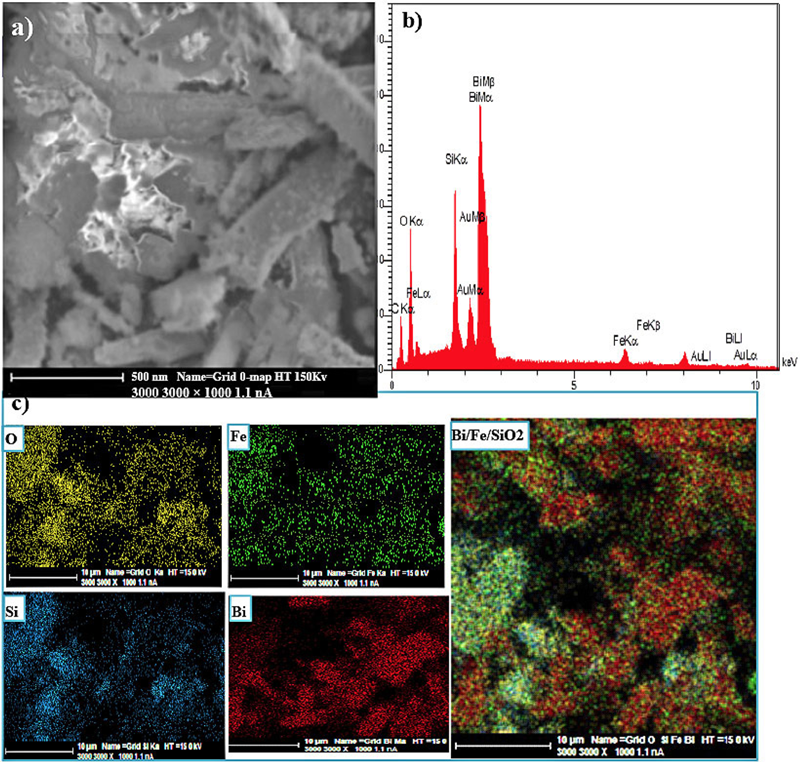
Figure 2
a)the SEM image of Nano-silica gel modified by iron and bismuth, b) the EDX image for Nano-silica gel modified by iron and bismuth, c) the Maping image for Nano-silica gel modified by iron and bismuth.
In order to review the structure of Nano-silica gel modified by iron and bismuth, Energy Dispersive X-ray Spectroscopy Analysis was used which specifies the elements forming of this Nano-adsorbent. As the information presented in figure 2b suggests, the iron and bismuth elements are visible. However, it is interesting to note that the same amount of these two elements was used at the beginning of the experiment but the spectrum suggests that there is a lower amount of iron than bismuth. This is because of the fact that the Energy Dispersive X-ray Spectroscopy analyzes the surface of the sample; therefore, it is possible that bismuth mostly exists on the surface and iron can be spotted in the lower layers but this analysis failed to specify this point. The maping analysis shows the amount of the elements on the surface. This indicates that these two elements can be spotted on the surface as shown in figure 2c. Figure 3 shows the TEM image. In this image, the diameter of the particles varies between 2 and 50 nanometers. The nanoparticles are shaped as a rod. The circular iron and bismuth nanoparticles can be seen within this rod. This image complies and overlaps with the SEM image in terms of appearance.

Figure 3
TEM image for Nano-silica gel modified by iron and bismuth.
3-3-Investigating the structure of adsorbents using XRD
The results of the XRD test have been presented in figures 4 and 5. The information presented in figure 4 is associated with the Nano-silica gel. By reviewing this information, it becomes totally clear that there is an amorphous structure, because there are no peaks that would show a symmetrical structure[(39)].
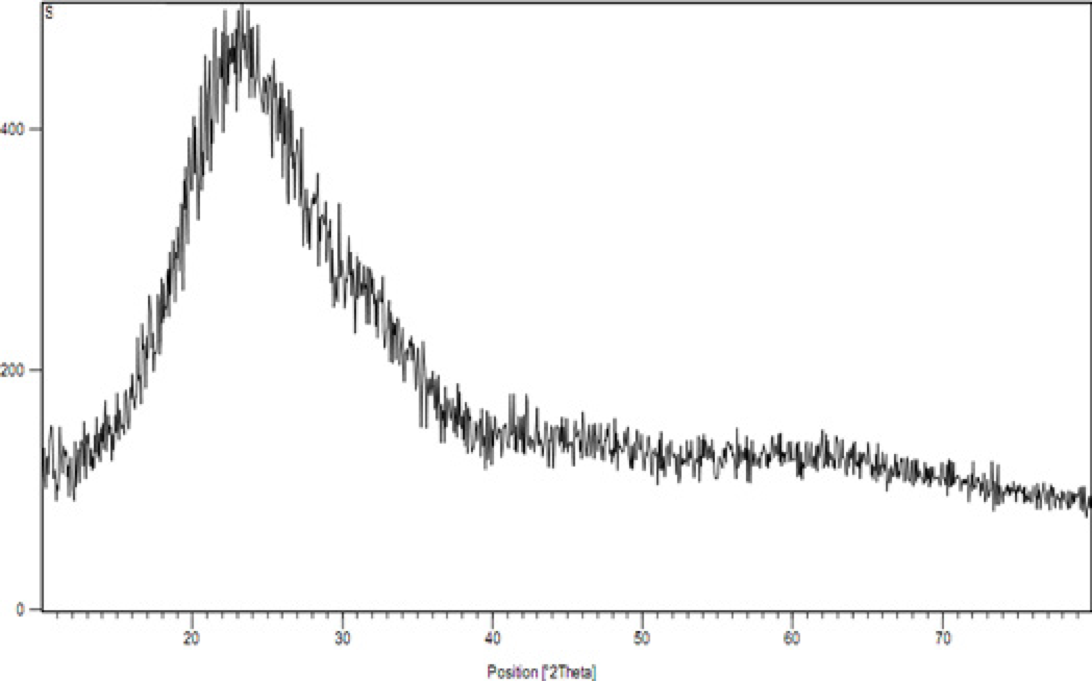
Figure 4
the XRD image for the Nano-silica gel adsorbent.
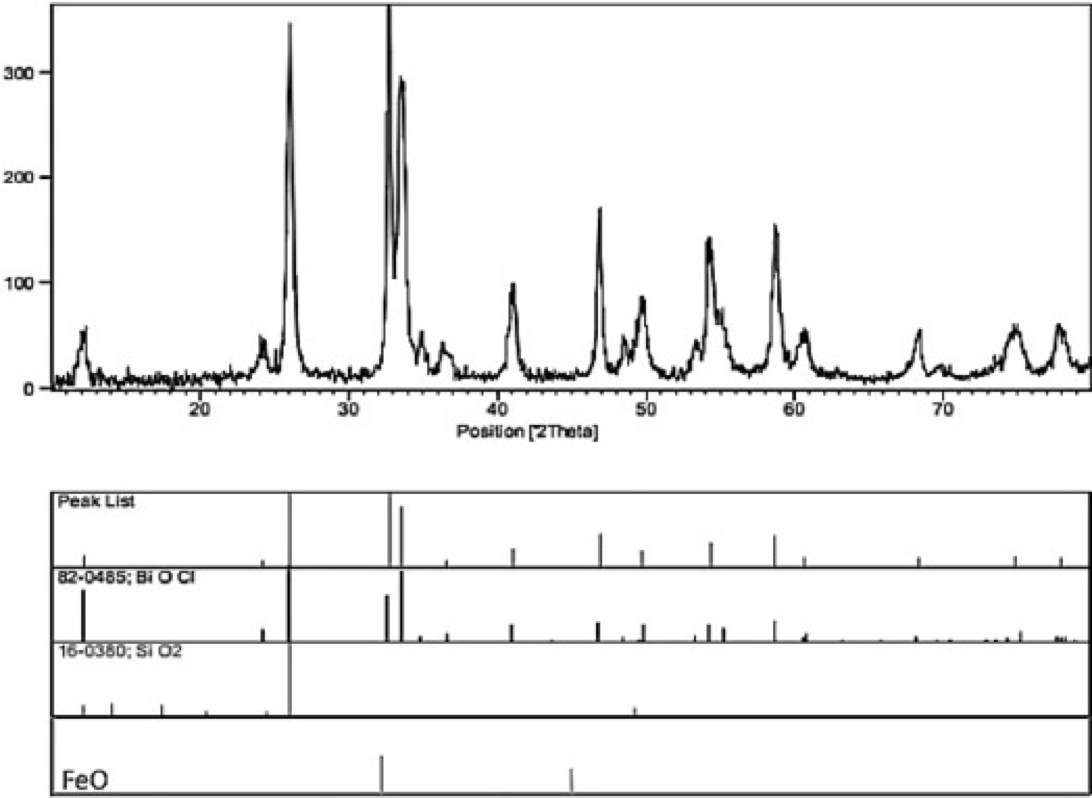
Figure 5
the XRD image for Nano-silica gel modified by iron and bismuth.
Zhang et al. (2008) also recorded one broadened XRD peak for amorphous silica centered at a 2θ value close to our measurement[(42)]. Martinez et al. (2006) prepared amorphous SiO2 by the sol-gel procedure, subjected it to thermal treatment, and then analyzed it using the Rietveld refinement. In their study, an amorphous peak was centered at 2θ = 23º[(43)]. The results obtained from the XRD test have been compared with the standard cards. They indicate that the principal peaks of the considered samples comply with the peaks of the SiO2 of the JCPDS card no. 16-0380 and for BiOCl the JCPDS card no. 82-0485 and this confirms the synthesis of this substance (figure 5). The first peak, which is at a significantly high point in the figure, shows the anisotropic crystallographic nature of the particles in this sample and the manner the particles are alongside one another. In this sample, no peak of impurity can be observed. In addition, by using the Scherrer equation associated with 28º [(101)], it can be concluded that the mean of the particle is approximately equal to 20 nm.
3-4-Investigating the effect of pH on the removal efficiency
PH plays a key role in the adsorption of the dye by adsorption. 0.08g of the adsorbent was added to 10mL of methylene blue with the concentration of 20 mg/L in order to evaluate the effect of pH on the percentage of adsorption and the pH was set between 2 and 11. NaOH and HCl were used to set the pH in an acidic or alkaline environments. Then, the sample was put on a shaker with a 200rpm speed for 20 minutes at room temperature. After that, the solution was placed in centrifuge at a 4000 rpm speed for 20 minutes and the percentage of removal of the dye was measured by the spectrophotometer. As the results presented in figure 6 indicate, the highest percentage of dye removal is related to the removal of methylene blue from the solution at pH=5 (98.02%) and pH=6 (98.00%). As the pH increases from 7 to 12, a decrease is seen in the percentage of adsorption as it falls to 92%. Similarly, a significant decline is seen in the percentage of adsorption in lower pHs (2-5) as it decreases to 90%.
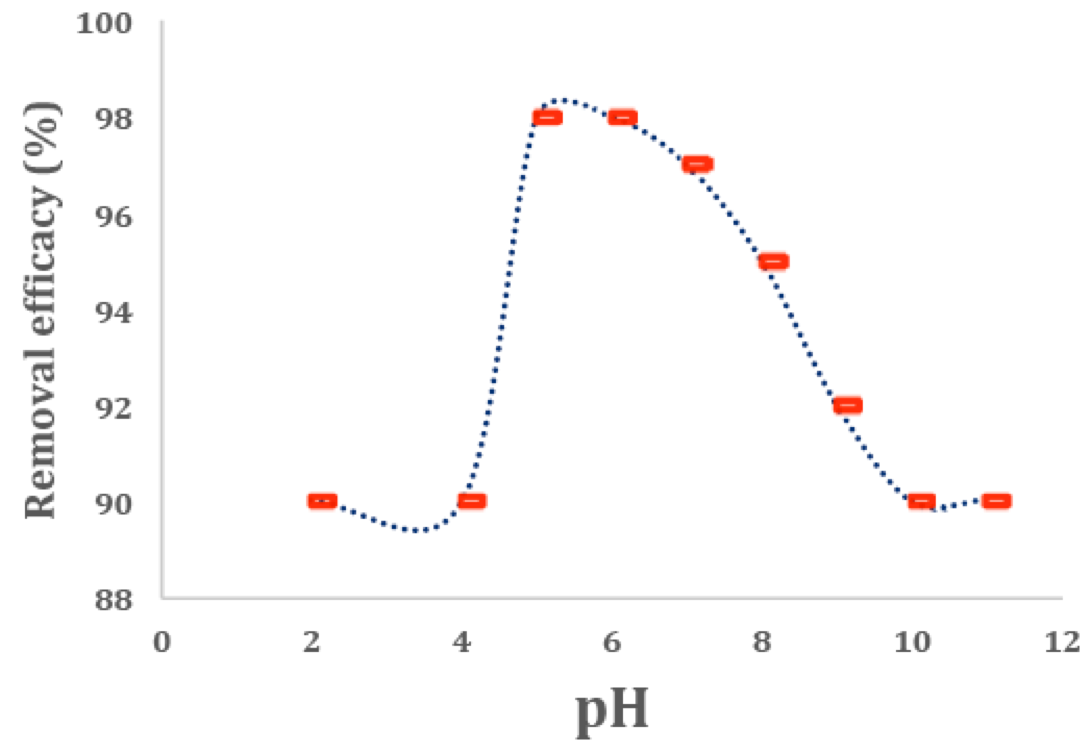
Figure 6
Effect of pH on adsorption of dye MB on the adsorbents (conditions: 8 mg adsorbent, 20 mg/L of 10 ml of cationic dyes duration of oscillation time of 20 min).
3-5-Investigating the effect of the amount of adsorbent on the removal efficiency
At this point, the effect of the amount of the adsorbent on the removal of the dye is reviewed. For this purpose, at the optimal pH (pH=6), various weights of the adsorbents were used for the methylene blue solution with the concentration of 20 mg/L. In each experiment, a contact was made between the adsorbent and 10 milliliters of the dye samples for 20 minutes. The results of this experiment have been presented in figure 7. As the results indicate, when the amount of the adsorbent increases from 0.01g to 0.1g, an increase is also seen in the percentage of removal of the dye. According to the experiments, the optimal mass for the adsorbent under study is 0.08g. Generally, as the mass of the adsorbent enhances, the adsorbent’s capacity to adsorb the dye increases too. Due to this fact, when the mass of the adsorbent increases, the specific surface area and the adsorption sites also experience an enhancement. In addition, as the amount of the adsorbent increase, no change is seen in the percentage of removal of the dye, since, the surface sites are filled and saturated [(44-46)].
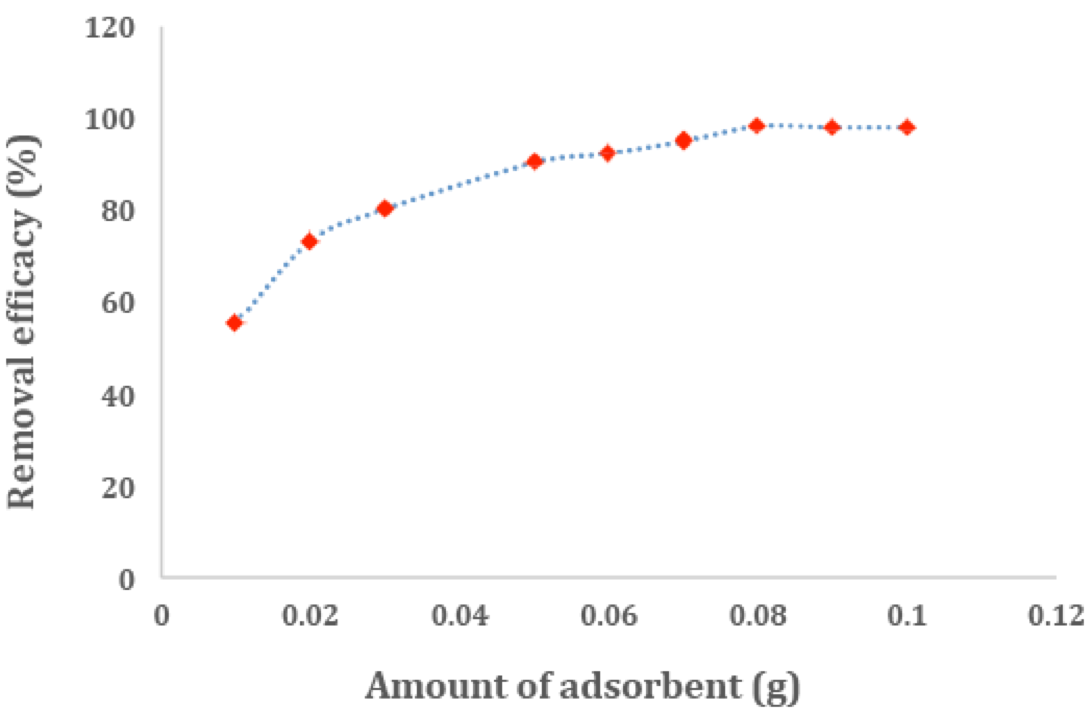
Figure 7
Variation curve of the percentage of removal of the dye in relation with the changes in the mass of the adsorbent
3-6- Investigating the effect of the time of performing on the reaction of the efficiency of removal
In order to perform this experiment, 10 samples were prepared. The concentration of the solution was 20 mg/L and the optimal pH for this experiment was 6. 0. 08g of the adsorbent was added to this solution and this experiment was performed in five-minute intervals (5-40 minutes) through which the optimal duration was calculated.
The results obtained from figure 8 show that as we increase the contact time, the removal would be more efficient and the most optimal removal has happened in the 20 minute where the percentage of removal reaches 98% and after that the increases in the percentage of removal efficiency have been insignificant. Since there are numerous empty sites on the surface of the adsorbent which are accessible, methylene blue in the aqueous solution is quickly adsorbed by the adsorbent in the first few minutes. As the contact time increases, the adsorption capacity enhances as well which is because of the fact when there is more time, the adsorbent and the adsorbate have more chances to make the adsorption better. Similar results were obtained by [(47, 48)]
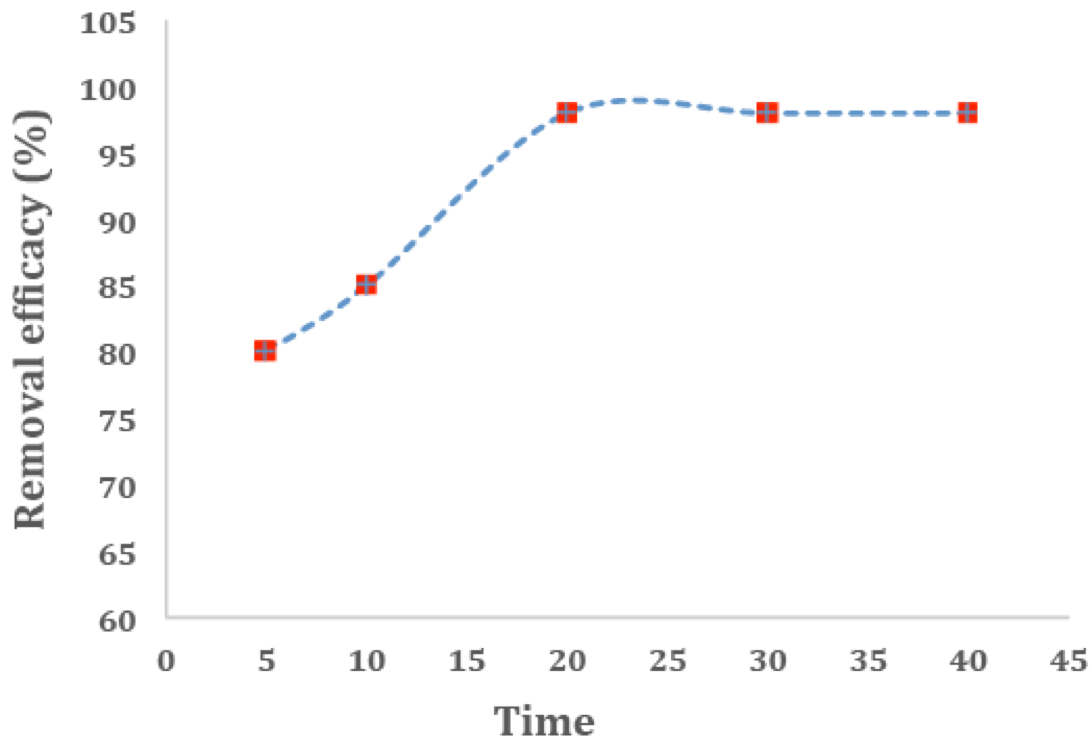
Figure 8
Effect of contact time of cationic dyes MB adsorption on adsorbents (conditions: 10 ml of 20 mg/L of dyes, pH = 6.0 and 80 mg adsorbent).
3-7-Investigating the effect of temperature of the reaction on the efficiency of the removal
Figure 9 illustrates the effect of the reaction on the adsorbent. By reviewing the temperature of the reaction, it became clear that the adsorption of methylene blue by modified Nano-SiO2 at a pH of 6 and 0.08g of the adsorbent and methylene blue with the concentration of 20 mg/L in 20 minutes at the room temperature (25 and 50ºC) increased. When the temperature is increased to 75 ºC, this amount will be reduced; therefore, given the fact that a 25 ºC temperature is easy to provide and no energy needs to be consumed, this is the best case. As the temperature rises, the percentage of dye removal decreases, since the methylene blue molecules move more and their adsorption by the surface reduces [(49)].
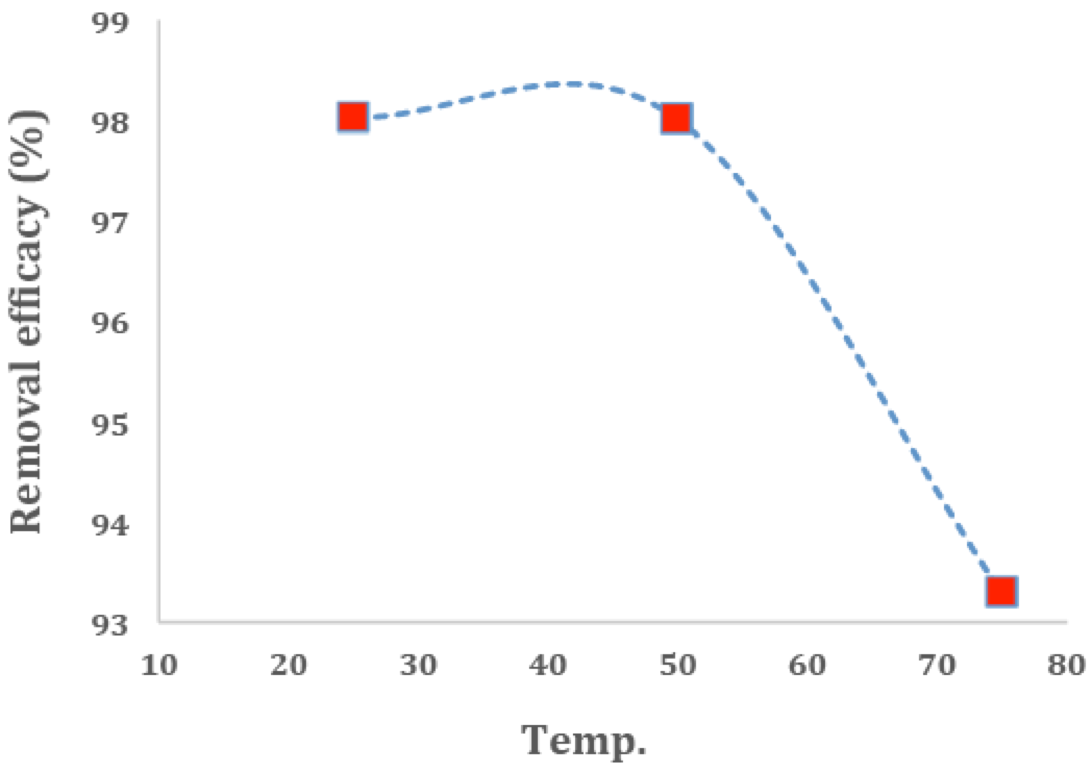
Figure 9
Effect of temperature of dye MB adsorption on adsorbents (conditions: 10 ml of 20 mg/L of dyes, pH = 6.0 and 80 mg adsorbent).
3-8-Investigating the effect of the concentration of the methylene blue solution on the efficiency of adsorption
Figure 10 shows the effect of the initial concentration of methylene blue on its surface adsorption by the adsorbent. In order to perform this experiment, the optimal time, the optimal mass of the adsorbent and the optimal pH were set (a mass of 0.08g, a 20-minute contact time and pH=6). As it can be seen in this figure, by increasing the initial concentration of methylene blue, the percentage of the adsorbed dye decreases in such a way that it can be argued that removal of the dye is affected by the initial concentration of it as well. At lower concentrations, methylene blue is adsorbed on the empty spots of the surface which are filled and saturated as the concentration increases. When the initial concentration of methylene blue is low, there are numerous accessible spots on the surface for the dye to be adsorbed. However, when the initial concentration of methylene blue enhances, the number of moles of methylene blue is very higher than the number of empty spots. Therefore, the accessible spots are quickly filled and the percentage of removal decreases. For instance, when the initial concentration increases from 30 to 80 mg/L, the percentage of adsorption of the dye by the adsorbent decreases from 98.00% to 50.00%.
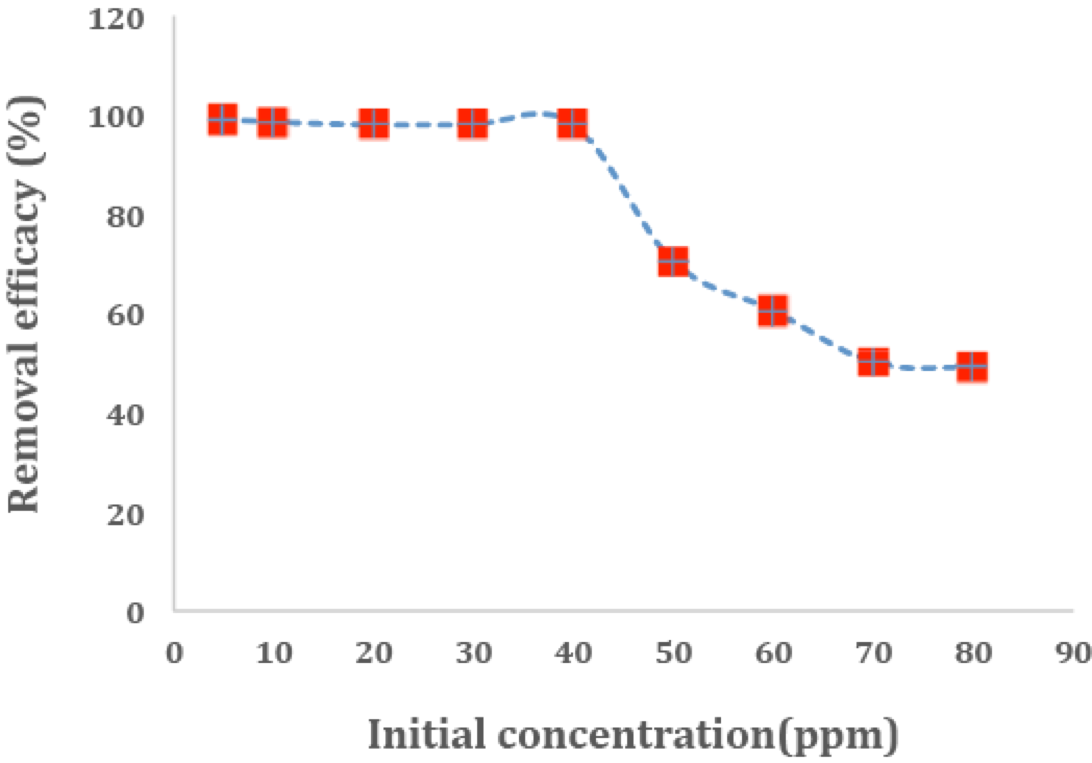
Figure 10
Effect of initial dye concentration on adsorption of MB (Initial dye concentration = 5-80 mg/L, contact time = 20 min, mass of adsorbent = 80 mg, solution volume =10 mL, pH = 6.0).
Experimental conditions remain unchanged; the different initial dye concentration of the adsorption is investigated. As shown in the Fig.8, the removal rate reduces with increasing concentration of MB [(50)]. For MB, the maximum adsorptions are 4.9 mg/g.
3-9-Comparing the amount of adsorption of methylene blue by the adsorbents like silica gel, Nano-silica gel and Nano-silica gel modified by iron and bismuth
By comparing these different adsorbents (silica gel, Nano-silica gel, and Nano-silica gel modified by iron and bismuth), the efficiency of the removal of methylene blue significantly increases if the Nano-silica gel modified by iron and bismuth is used as the adsorbent.
In brief, the purposes behind using Nano-silica gel modified by iron and bismuth are:
-
Increasing the amount of adsorption of methylene blue
-
Decreasing the time of reaction
-
Decreasing the amount of the adsorbent
-
The information presented in table 1 displays the results of removing methylene blue from the solution.
-
Amount of adsorption: Nano-silica gel modified by iron and bismuth > Nano-silica gel > silica gel
Effect of different adsorbent on adsorption of MB (Initial dye concentration = 20 mg/L, contact time = 20 min, mass of adsorbent = 80 mg, solution volume =10 mL, pH = 6.0)

3-10-Investigating the equilibrium isotherms
In this study, Langmuir and Freunlich isotherm models have been used for the surface adsorption of methylene blue on modified silica gel which have been presented in figure 11. Moreover, all of these results have been presented in table 1.
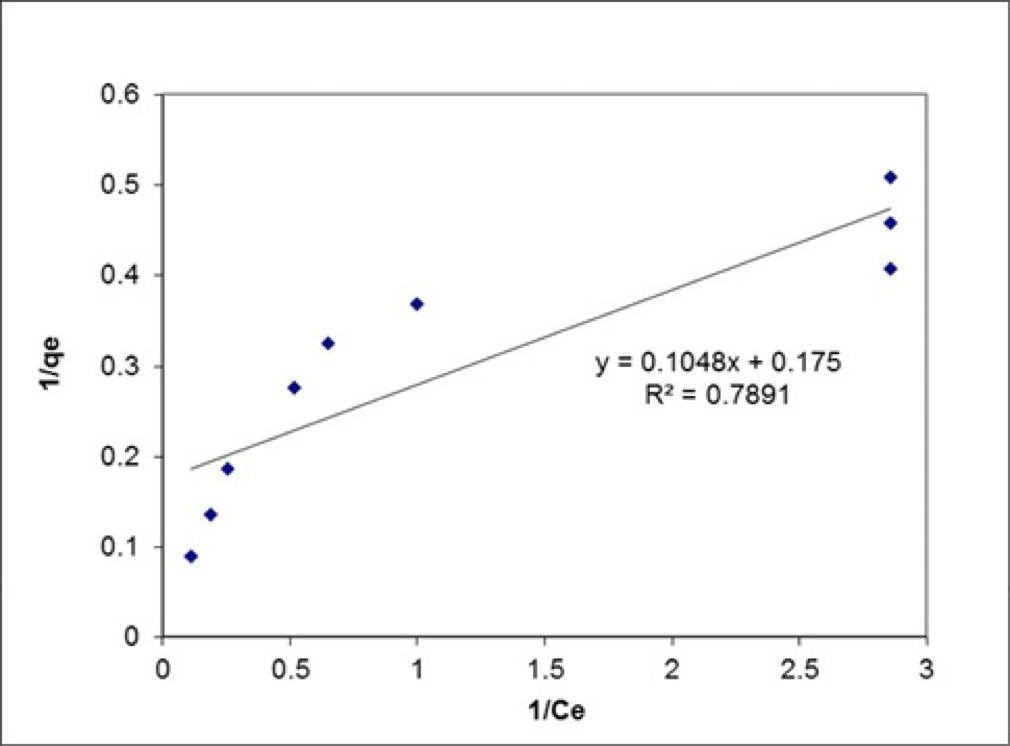
Figure 11
Freundlich and Langmuir isotherm for the adsorption of methylene blue on modified silica gel.
It is important to study equilibrium since we might grasp significant information about the nature of the interaction between the adsorbate and the adsorbent which can be used for determining the adsorbent’s adsorption capacity. There are different models for describing the data obtained from the experiments and the most important model is adsorption isotherms. In this work, Langmuir and Freunlich isotherm models have been used for reviewing the adsorption process. The Langmuir isotherm model is based on the assumption that a one-layered cover of the adsorbents are placed on the adsorbent’s uniform surface structure and there is no interaction between the adsorbent molecules and the adsorption sites which are similar. In addition, when the adsorbed molecules are adsorbed on the surface, they should not be separated[(45)]. The adsorption equation is as follows:
Where Ce (mg L-1) is the equilibrium concentration, qe (mg g-1) is the amount of dye adsorbed at equilibrium, qmax (mg g-1) is the maximum adsorption capacity of the absorbents, and KL is the Langmuir adsorption constant. Equilibrium parameter RL is used to determine whether the adsorption is favorable. It can be defined in the following equation:
Where C0 (mg L-1) is the initial dye concentration and KL (L mg-1) is the Langmuir constant related to the energy of the adsorption. The values of RL for dyes lie between 0 and 1 [(51)], thereby confirming that the adsorption is a favorable process. If the adsorption process can be described by the equation, it indicates that the process is single molecule layer adsorption.
Freundlich isotherm equation is based on the assumption that adsorption happens on an inhomogeneous surface and the energy of the adsorption process has a non-uniform distribution on the surface of the adsorbent. It is because of this inhomogeneity that there are various factor groups on the surface and there are different mechanisms for the interaction between the adsorbent and the adsorbate[(45)]. This isotherm model is shown as follows:
Where qe (mg g-1) is the amount of dye adsorbed on the adsorbents, Kf is an approximate indicator of adsorption capacity. The value of 1/n between 0 and 1 indicates favorable adsorption process. If the value of 1/n ≥ 2, this indicates that the adsorption process is difficult to carry out.
The data obtained from the studies focused on the adsorption equations and their results have been presented in table 2. Since the Freundlich model has a higher correlation (0.912) than the Langmuir model (R=0.789), it is better to use it in order to describe the adsorption equilibrium. The maximum adsorption capacity was calculated by theoretical. The obtained value is similar to the experimental data value.
Isotherm parameters for the adsorption of methylene blue on modified silica gel.
In addition, the obtained value of 1/n=0.454 corresponds to a favorable adsorption process. On the other hand, the RL values of MB obtained from Langmuir isotherm lied 0.0769 which indicated that the adsorption was favorable, in consistence with the Freundlich model[(52)]
3-11-Kinetic studies
In order to make a compliance between the empirical data associated with the surface adsorption of the contaminants on the adsorbent, different kinetic models have been used such as pseudo-first-order kinetic model and pseudo-second-order kinetic model. A kinetic model is one of the most important factors for designing an adsorption system (in order to determine the optimal contact time), predicting the speed and process of adsorption which is controlled by the kinetics. The linearized pseudo first and second order are generally expressed in the following table[(53, 54)].
The information presented in this table depicts how the contact time affects the adsorption process at different times. It must be noted that all of the tests and experiments have been performed at ambient temperature. The adsorption behavior showed that the adsorption system best fit with the pseudo-second-order (R2=0.999) model which illustrates that the rate-limiting step can be chemical adsorption or chemisorption involving valence forces through sharing or exchange of electrons between adsorbent and adsorbate[(55)]. In the pseudo-first order model, K1 is equal to 0.0403 and in the pseudo-second order model, K2=0.263 (figure 12).
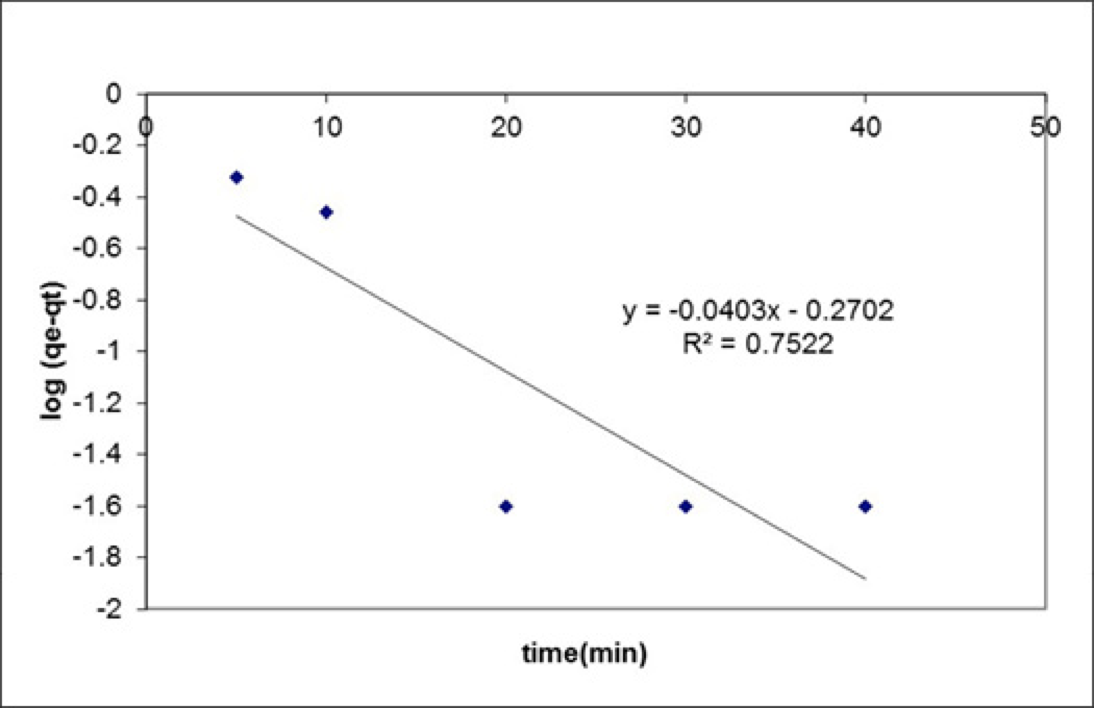
Figure 12
Pseudo-first-order and pseudo-second-order adsorption kinetics for the adsorption of methylene blue on modified silica gel as the adsorbent.
Lagergren pseudo-first order kinetic and pseudo-second order kinetic data.
4. Conclusion
We have prepared modified nano-silica with Bismuth and Iron for adsorption of methylene blue solution. SEM, TEM, EDX, XRD and FTIR were used to study the structure of the adsorbents. The key parameters such as pH, contact time, amount of adsorbent and initial concentration of methylene blue on adsorption treatment were studied. Results indicated that modified nano-silica by iron and bismuth as an adsorbent is known as a novel remover of methylene blue. The experiments were performed with the purpose of removing methylene blue from a dye solution by adsorption. Based on 20 mg/L MB as initial concentration, the optimal pH is 5 to 6, the optimal contact time is 20 minutes and the optimal amount of the adsorbent is 8gr/L. The kinetics and isotherm adsorption data were well described by the pseudo-second-order model and the Freundlich isotherm model, respectively.
Reference
1.. 1. Brookstein, D.S., Dermatologic clinics, 2009, 27, 309-322.
2.. 2. de Lima, R.O.A.;Bazo, A.P.;Salvadori, D.M.F.;Rech, C.M.;de Palma Oliveira, D., de Aragão Umbuzeiro, G., Mutation Research/Genetic Toxicology and Environmental Mutagenesis, 2007, 626, 53-60.
3.. 3. Royer, B.;Cardoso, N.F.;Lima, E.C.;Vaghetti, J.C.;Simon, N.M.;Calvete, T., Veses, R.C., Journal of Hazardous Materials, 2009, 164, 1213-1222.
4.. 4. Ponnusami, V.;Madhuram, R.;Krithika, V., Srivastava, S., Chemical Engineering Journal, 2008, 140, 609-613.
5.. 5. Rafatullah, M.;Sulaiman, O.;Hashim, R., Ahmad, A., Journal of Hazardous Materials , 2010, 177, 70-80.
6.. 6. Bratby, J., Water Intelligence Online, 2006, 5, 9781780402321.
7.. 7. Vogelpohl, A., Kim, S.-M., Journal of Industrial and Engineering Chemistry, 2004, 10, 33-40.
8.. 8. Ma, D.-Y.;Wang, X.-H.;Song, C.;Wang, S.-G.;Fan, M.-H., Li, X.-M., Desalination, 2011, 276, 233-238.
9.. 9. Verma, A.K.;Dash, R.R., Bhunia, P., Journal of Environmental Management, 2012, 93, 154-168.
10.. 10. Lima, E.C.;Royer, B.;Vaghetti, J.C.;Simon, N.M.;da Cunha, B.M.;Pavan, F.A.;Benvenutti, E.V.;Cataluña-Veses, R., Airoldi, C., Journal of Hazardous Materials , 2008, 155, 536-550.
11.. 11. Kalaitzidou, K.;Fukushima, H.;Askeland, P., Drzal, L.T., Journal of materials science, 2008, 43, 2895-2907.
12.. 12. Zhuravlev, L., Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2000, 173, 1-38.
13.. 13. Iler, R., JohnWiley and Sons, New York, 1979.
14.. 14. Zuyi, T.;Taiwei, C.;Weijuan, L.;Jinzhou, D.;Xiongxin, D., Yingjie, G., Colloids and Surfaces A: Physicochemical and Engineering Aspects , 2004, 242, 39-45.
15.. 15. Anbia, M.;Hariri, S.A., Ashrafizadeh, S., Applied Surface Science, 2010, 256, 3228-3233.
16.. 16. Lim, S.-H.;Woo, E.-J.;Lee, H., Lee, C.-H., Applied Catalysis B: Environmental, 2008, 85, 71-76.
17.. 17. Deligeer, W.;Gao, Y., Asuha, S., Applied Surface Science , 2011, 257, 3524-3528.
18.. 18. Emadi, M.;Shams, E., Amini, M.K., Journal of Chemistry, 2012, 2013.
19.. 19. Fan, F.-L.;Qin, Z.;Bai, J.;Rong, W.-D.;Fan, F.-Y.;Tian, W.;Wu, X.-L.;Wang, Y., Zhao, L., Journal of environmental radioactivity, 2012, 106, 40-46.
20.. 20. Sheikhian, L., Desalination and Water Treatment, 2016, 57, 8447-8453.
21.. 21. Han, H.;Wei, W.;Jiang, Z.;Lu, J.;Zhu, J., Xie, J., Colloids and Surfaces A: Physicochemical and Engineering Aspects , 2016, 509. 539-549.
22.. 22. Ma, K.;Sai, H., Wiesner, U., Journal of the American Chemical Society, 2012, 134, 13180-13183.
23.. 23. Reddy, K.R.;Lee, K.-P.;Gopalan, A.I., Kang, H.-D., Reactive and Functional Polymers, 2007, 67, 943-954.
24.. 24. Zhang, Y.P.;Lee, S.H.; Reddy, K.R. ; Gopalan, A.I. , Lee, K.P., Journal of Applied Polymer Science, 2007, 104, 2743-2750.
25.. 25. Gao, J.; Wei, W. ;Shi, M.; Han, H. ; Lu, J. , Xie, J. , Journal of materials science , 2016, 51, 4481-4494.
26.. 26. Bulut, Y., Aydın, H., Desalination , 2006, 194, 259-267.
27.. 27. Kavitha, D., Namasivayam, C., Bioresource Technology, 2007, 98, 14-21.
28.. 28. Han, R.;Zhang, J.;Han, P.; Wang, Y. ;Zhao, Z., Tang, M., Chemical Engineering Journal , 2009, 145, 496-504.
29.. 29. Jain, R.;Mathur, M.;Sikarwar, S., Mittal, A., Journal of Environmental Management , 2007, 85, 956-964.
30.. 30. Selvam, P.P.;Preethi, S.;Basakaralingam, P.;Thinakaran, N.;Sivasamy, A., Sivanesan, S., Journal of Hazardous Materials , 2008, 155, 39-44.
31.. 31. Das, S.K.;Bhowal, J.;Das, A.R., Guha, A.K., Langmuir, 2006, 22, 7265-7272.
32.. 32. Peng, L.;Qin, P.;Lei, M.;Zeng, Q.;Song, H.;Yang, J.;Shao, J.;Liao, B., Gu, J., Journal of Hazardous Materials , 2012, 209, 193-198.
33.. 33. Yousefi, M., Karami, C., Advances in Environmental Biology, 2014, 2113-2117.
34.. 34. Toshima, N., Yonezawa, T., New J. Chem., 1998, 22, 1179-1201.
35.. 35. Kowshik, M.;Ashtaputre, S.;Kharrazi, S.;Vogel, W.;Urban, J.;Kulkarni, S.K., Paknikar, K., Nanotechnology, 2003, 14, 95.
36.. 36. Hayashi, H.;Sugiyama, S.;Shigemoto, N.;Miyaura, K.;Tsujino, S.;Kawashiro, K., Uemura, S., Catal. Lett., 1993, 19, 369-373.
37.. 37. Karami, C.;Ahmadian, H.;Nouri, M.;Jamshidi, F.;Mohammadi, H.;Ghodrati, K., Hamidi, Z., Catal. Commun., 2012, 27, 92-96.
38.. 38. Mirzaei, A.A.;Faizi, M., Habibpour, R., Applied Catalysis A: General, 2006, 306, 98-107.
39.. 39. Musić, S.;Filipović-Vinceković, N., Sekovanić, L., Brazilian Journal of Chemical Engineering, 2011, 28, 89-94.
40.. 40. Wang, Y. ;Zhao, J.;Zhao, X.;Tang, L.;Li, Y., Wang, Z., Materials Research Bulletin, 2009, 44, 220-223.
41.. 41. Kassaee, M.;Masrouri, H., Movahedi, F., Applied Catalysis A: General , 2011, 395, 28-33.
42.. 42. Zhang, G.;Xu, Y.;Xu, D.;Wang, D.;Xue, Y., Su, W., High Pressure Research, 2008, 28, 641-650.
43.. 43. Martínez, J.;Palomares-Sánchez, S.;Ortega-Zarzosa, G.;Ruiz, F., Chumakov, Y., Materials letters, 2006, 60, 3526-3529.
44.. 44. Ghaedi, M.;Nasab, A.G.;Khodadoust, S.;Rajabi, M., Azizian, S., Journal of Industrial and Engineering Chemistry , 2014, 20, 2317-2324.
45.. 45. Guo, J.-Z.;Li, B.;Liu, L., Lv, K., Chemosphere, 2014, 111, 225-231.
46.. 46. Marković, S.;Stanković, A.;Lopičić, Z.;Lazarević, S.;Stojanović, M., Uskoković, D., Journal of Environmental Chemical Engineering, 2015, 3, 716-724.
47.. 47. Cengiz, S., Cavas, L., Bioresource Technology , 2008, 99, 2357-2363.
48.. 48. Yan, B.;Chen, Z.;Cai, L.;Chen, Z.;Fu, J., Xu, Q., Applied Surface Science , 2015, 356, 39-47.
49.. 49. Pal, S.;Ghorai, S.;Das, C.;Samrat, S.;Ghosh, A., Panda, A.B., Industrial & Engineering Chemistry Research, 2012, 51, 15546-15556.
50.. 50. Hii, S.-L.;Yong, S.-Y., Wong, C.-L., Journal of applied phycology, 2009, 21, 625-631.
51.. 51. Fayazi, M.;Ghanei-Motlagh, M., Taher, M.A., Materials Science in Semiconductor Processing, 2015, 40, 35-43.
52.. 52. Danesh, N.;Hosseini, M.;Ghorbani, M., Marjani, A., Synthetic Metals, 2016, 220, 508-523.
53.. 53. Meena, A.K.;Mishra, G.;Rai, P.;Rajagopal, C., Nagar, P., Journal of Hazardous Materials , 2005, 122, 161-170.
54.. 54. Hamzeh, Y.;Ashori, A.;Azadeh, E., Abdulkhani, A., Materials Science and Engineering: C, 2012, 32, 1394-1400.
55.. 55. Ciopec, M.;Davidescu, C.;Negrea, A.;Grozav, I.;Lupa, L.;Negrea, P., Popa, A., Chemical engineering research and design, 2012, 90, 1660-1670.
Author notes
*Corresponding author: *E-mail: f.salimi@iauksh.ac.ir (F.Salimi), Tel: 0098-341-3221452
