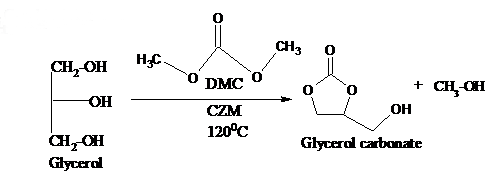
A facile synthesis of glycerol carbonate by transesterification of glycerol with dimethyl carbonate: Ceria based catalysts
DOI:
https://doi.org/10.29356/jmcs.v62i4.859Keywords:
Glycerol, Ceria/Zirconia/Magnesia, Glycerol carbonate, TransesterificationAbstract
Abstract. Solid base catalysts such as Ceria-Zirconia-Magnesia with different mole ratio of magnesium were prepared by impregnation method and characterized by CO2-TPD, PXRD, FT-IR and ICP-OES analysis. The catalytic activity of the catalysts was tested in the liquid phase transesterification of glycerol with dimethyl carbonate to synthesise glycerol carbonate. Optimization of reaction condition was carried out by varying the molar ratio of the reactants, temperature and time. The highest yield (97 %) of glycerol carbonate was obtained at a reactant molar ratio of 1:3 at 120 °C in 6 h. Study of reusability and reactivation of solid base catalyst was also taken up. A suitable base catalysed mechanism for the formation of glycerol carbonate is proposed.
Resumen. Se prepararon catalizadores de base sólida como Ceria-Zirconia-Magnesia con diferentes proporciones molares de magnesio mediante el método de impregnación y se caracterizaron por análisis de CO2-TPD, PXRD, FT-IR e ICP-OES. La actividad catalítica de los catalizadores se probó en la transesterificación en fase líquida de glicerol con carbonato de dimetilo para sintetizar carbonato de glicerol. La optimización de las condiciones de reacción se llevó a cabo variando la relación molar de los reactivos, la temperatura y el tiempo. El mayor rendimiento (97 %) de carbonato de glicerol se obtuvo a una relación molar reactiva de 1:3 a 120 °C en 6 h. También se realizó un estudio de la reutilización y reactivación del catalizador de base sólida. Se propone un mecanismo catalítico básico adecuado para la formación de carbonato de glicerol.
Downloads
References
Marakatti, S, V.; Anand Halgeri B.; RSC Adv, 2015, 5, 14286-14293.
Tanb, Y. Z.; Liu, J.; Wang, Kang, M.; Yin, N.; Wang, X.; Zhub, Y.; J. Braz. Chem. Soc, 2014, 25, 152-160.
Burk, R. M.; Roof, M. B.; Tetrahedron Lett, 1993, 34, 395-398.
Mizuno, T.; Nakai, T.; Mihara, M.; Heteroat. Chem, 2010, 21, 99-102.
Hu, J.; Li, J.; Gu, Y.; Guan, Z.; Ni, W.; Li, Y.; Li, T.; Appl. Catal. A: Gen, 2010, 386, 188- 193.
Vieville, C.; Yoo, J.; Pelet, S.; Mouloungui, Z.; Catal. Lett, 1998, 56, 245-247.
George, J.; Patel, Y.; Pillai, S.M.; Munshi, P.; J. Mol. Catal. A: Chem, 2009, 304, 1-7.
Climent, M.J.; Corma, A.; Frutos, P. D.; Iborra, S.; Noy, M.; Velty, A.; J. Catal, 2010, 269, 140-149.
Hammond, C.; Lopez-Sanchez, J. A.; Rahim, M. H. A; Dimitratos, N.; Jenkins, R. L.; Carley, A. F.; He, Q.; Kiely C. J.; Knight, D.W.; Hutchings, G.J.; Dalton Trans, 2011, 40, 3927-3937
Ochoa-Gómez, J.R; Gómez-Jiménez-Aberasturi, O.; Maestro Madurga, B.; Pesquera- Rodríguez A.; Ramírez López, A. C.; Lorenzo-Ibarreta L.; Torrecilla-Soria, J.; Villarán-Velasco, M. C.; Appl. Catal. A: Gen, 2009, 366, 315-324.
Bell, J. B.; US Pat, 1959, 2, 915 -529.
Alvarez, M. G.; Segarra, A. M.; Contreras, S.; Sueiras, J. E.; Medina, F.; Figueras, F.; Chem. Eng. J, 2010, 161, 340-345.
Bai, R.; Wang, Y.; Wang, S.; Mei, T.; Li, G.; Li, Fuel Process. Technol, 2010, 106, 209-214.
Bancquart, S.; Vanhove, C.; Pouilloux, Y.; Barrault, J.; J. Appl. Catal. A: Gen, 2001,218, 1-11
Sato, S.; Sato, F.; Gotoh, H.; Yamada, Y.; ACS Catal, 2013, 3, 721–734.
Venkatesh, Mohammed Shamshuddin, S. Z.; Shyamsundar, M.; Vasanth, V. T.; J. Mex. Chem. Soc, 2014, 58, 378-385.
Gotoh, H.; Yamada, Y.; Sato, S.; Appl. Catal. A: Gen, 2010, 377, 92-98.
Sakata, Y.; Ponec, V.; Appl. Catal. A: Gen, 1998, 166, 173-184.
Kobune, M.; Sato, S.; Takahashi, R.; J. Mol. Catal. A: Chem,2008, 279, 10-19.
Rathod, S.; Arbad, B.; Lande, M.; Chinese J of Catal, 2010, 31, 631-636.
Trovarelli, A.; catalysis by ceria and related materials London, 2013, 12, 1-888.
Rathod, B. S.; Machhinndra, K.; Lande, R.; Balasaheb, Arbad, Gambhire, A.; Arabian J of Chem, 2014, 7, 373-378.
Hussein, A.; Khalaf, Springer plus, 2013, 2:619, 1-9.
Parameswaram, G.; Srinivas, M.; HariBabu, B.; Sai Prasad, P. S.; Lingaiah, N.; Catal. Sci. Technol, 2013, 3, 3242-3249.
Malyaadri, M.; Jagadeeswaraiah, K.; Sai Prasad P. S.; Lingaiah, N.; Catalysts. Appl.Catal. A: Gen, 2011, 401, 153-157.
Peterson Santos Querino, Jose Renato Carlos Bispo, Maria do Carmo Rangel, Catalysis Today, 2005, 107, 930-925.
Rathoda, S.; Navgireb, M.; Arbadb, B.; Lande, M. S.; Afr. J. Chem, 2012, 65, 196-201.
Hattori H.; Chem Rev, 1995, 95, 537-558.
Tsuji H.; Hattori H.; Catal Today, 2006, 116, 239-243.
Okoye, P. U.; Abdullah, Z. B.; Hameed, H.; J. Taiwan Institute of Chem. Eng, 2016, 68, 51-58.
Zheng, L.; Xia, S.; Lu, X. Z.; Hou, Z.; Chinese. J. Catal, 2015, 36, 1759-1765.
Khayoon, M.S.; Hameed, B.H.; Appl Catal A Gen, 2013, 466, 272-281.
Prakruthi, H. R.; Jai Prakash, B. S.; Bhat, Y. S.; J. Mol. Catal. A: Chem, 2015, 408, 214-220.
Liu, J.; Li, Y.; Zhang, J.; He, D. App Catal A: Gen, 2016, 513. 9-18.
Cinzia Chiappe.: Sunita Rajamani: Pure Appl. Chem, 2012, 84, 755–762.
Yin Wang: Chunling Liu: Jihong Sun: Rongzhen Yang: Wensheng Dong.: Sci. china chem., 2015, 58, 708–71.
Jose, R.; Gomez, C.; Go, O. Aberasturi, M. N. J.; Madurga, B. M.; Rodr?guez, A. P.; Lopez, C. R.; Ibarreta, L. L.; Soria, J. T.; Velasco, C. M. V.; App Catal A: Gen, 2009, 366, 315-324.
Downloads
Published
Issue
Section
License
Authors who publish with this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgement of its initial publication in this journal.